Researchers from Dr. Nicolas Kaeffer’s Organometallic Electrocatalysis team in the Molecular Catalysis department of Prof. Walter Leitner at MPI CEC have disclosed that the hydrogenation of alkynes to olefins can be performed using electricity and bypassing hydrogen gas, thanks to a nickel-based catalyst. The investigated transformation - the partial hydrogenation of alkynes to olefins - is widely used in organic synthesis.
This electrocatalytic approach fits into the strategy of steering away from fossil fuels as the main source of goods, fuels and materials. The ultimate goal is to combine electricity from renewable sources with sustainable raw materials in an electrochemical approach for the production of chemicals.
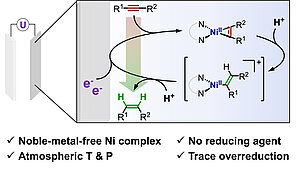
In the chemical value chain, olefins are among the most important precursors for base and fine chemicals (as for instance in polymer chemistry), and are widely obtained from their parent alkynes using hydrogen gas. A major challenge in this transformation is the over-hydrogenation of the starting alkyne that results in alkane compounds of lower added-value.
In this study, the scientists show that the production of olefins from alkyne compounds is electrochemically catalyzed by a simple molecular nickel complex in a facile manner at ambient temperature and pressure. Moreover, this system exhibits little to no undesirable over-hydrogenation to alkanes. By coupling electrochemical analysis and spectroscopy, it was also found that the catalytic cycle is initiated in an atypical way by activation of the alkyne compound, rather than by the formation of a hydride as is usually the case.
As a proof-of-concept for molecular electrocatalytic alkyne semihydrogenation, this work opens a platform to develop original hydrogenations and functionalizations of alkynes via their electrochemical activation.
Original Paper: Mi-Young Lee, Christian Kahl, Nicolas Kaeffer, and Walter Leitner (2022). Electrocatalytic Semihydrogenation of Alkynes with [Ni(bpy)3]2+. JACS Au 2022, https://doi.org/10.1021/jacsau.1c00574