Prof. Dr. Serena DeBeer - Inorganic Spectroscopy

- Prof. Dr. Serena DeBeer
- Director
- Inorganic Spectroscopy
- +49 (0)208 306 - 3656
- serena.debeer(at)cec.mpg.de
- Room: 608
Vita
| B.S. (Chemistry) | Southwestern University, TX, USA (1995) |
| Ph.D. (Chemistry) | Stanford University, CA, USA (2002) |
| Beam Line Scientist | SSRL, SLAC, Stanford, University (2001-2003) |
| Staff Scientist | SSRL, SLAC, Stanford, University (2003-2009) |
| Assistant Professor | Cornell University, NY, USA (2009-2012) |
| Research Group Leader | MPI for Bioinorganic Chemistry; today: MPI CEC (2011-2017) |
| Group leader | PINK Beamline, Energy Materials In-Situ Laboratory, Helmholtz Zentrum, Berlin (since 2012) |
| Adjunct Professor | Department of Chemistry and Chemical Biology, Cornell University (since 2012) |
| Honorary Faculty | Ruhr University Bochum (since 2014) |
| Director | Inorganic Spectroscopy, MPI CEC (since 2017) |
| Honorary Faculty | University of Duisburg-Essen (since 2024) |
Publications
Full publications list | ORCID | ResearcherID | Google Scholar Profile
Newest publications
2025
Fang, W., Zhang, Y., Kang, L., DeBeer, S., Leitner, W., Bordet, A., Risager, A. (2025). Molecularly modified aluminum phosphates as support materials for Ru nanoparticles in selective hydrogenation. Journal of Catalysis, 442, 115911. doi:10.1016/j.jcat.2024.115911.
Sengupta, K., Joyce, J. P., Decamps, L., Kang, L. Q., Björnsson, R., Rüdiger, O., DeBeer, S. (2025). Investigating the Molybdenum Nitrogenase Mechanistic Cycle Using Spectroelectrochemistry. Journal of the American Chemical Society. doi:10.1021/jacs.4c16047.
McCubbin Stephanic, O., Pollock, C. J., Zielinski, K. A., Foschi, W., Rice, D. B., Lois, P., DeBeer, S. (2025). Implementation of simultaneous ultraviolet/visible and x-ray absorption spectroscopy with microfluidics. Review of Scientific Instruments, 96 (1) 015107. doi:10.1063/5.0218572.
2024
Zhang, Y. Y., Levin, N., Kang, L. Q., Müller, F., Zobel, M., DeBeer, S., Leitner, W., Bordet, A. (2024). Design and Understanding of Adaptive Hydrogenation Catalysts Triggered by the H2CO2-Formic Acid Equilibrium. Journal of the American Chemical Society, 146 (44), 30057-30067. doi:10.1021/jacs.4c06765.
Zhang, B. F., Joyce, J. P., Wolford, N. J., Brennessel, W. W., DeBeer, S., Neidig, M. L. (2024). Unusual S=1 Four-Coordinate Fe(IV) Complexes Supported by Bisamide Ligands: Syntheses, Characterization, and Electronic Structures. Angewandte Chemie-International Edition, 63 (36). doi:10.1002/anie.202405113.
Zenner, J., Tran, K., Kang, L. Q., Kinzel, N. W., Werle, C., DeBeer, S., Bordet, A., Leitner, W. (2024). Synthesis, Characterization, and Catalytic Application of Colloidal and Supported Manganese Nanoparticles. Chemistry-a European Journal, 30 (25). doi:10.1002/chem.202304228.
Wilson, D. W. N., Fataftah, M. S., Mathe, Z., Mercado, B. Q., DeBeer, S., Holland, P. L. (2024). Three-Coordinate Nickel and Metal-Metal Interactions in a Heterometallic Iron-Sulfur Cluster. Journal of the American Chemical Society, 146 (6), 4013-4025. doi:10.1021/jacs.3c12157.
Wahl, I. M., Sengupta, K., van Gastel, M., Decamps, L., DeBeer, S. (2024). Understanding the P-Cluster of Vanadium Nitrogenase: an EPR and XAS Study of the Holo vs. Apo Forms of the Enzyme. ChemBioChem. e202400833. doi:10.1002/cbic.202400833.
Van Stappen, C., Van Kuiken, B. E., Mörtel, M., Ruotsalainen, K. O., Maganas, D., Khusniyarov, M. M., DeBeer, S. (2024). Correlating Valence and 2p3d RIXS Spectroscopies: A Ligand-Field Study of Spin-Crossover Iron(II). Inorganic Chemistry, 63 (16), 7386-7400. doi:10.1021/acs.inorgchem.4c00435.
Tamhankar, A., Wensien, M., Jannuzzi, S. A. V., Chatterjee, S., Lassalle-Kaiser, B., Tittmann, K., DeBeer, S. (2024). In Solution Identification of the Lysine-Cysteine Redox Switch with a NOS Bridge in Transaldolase by Sulfur K-Edge X-ray Absorption Spectroscopy. Journal of Physical Chemistry Letters, 15 (16), 4263-4267. doi:10.1021/acs.jpclett.4c00484.
Tagliavini, V., Duan, P. C., Chatterjee, S., Ferretti, E., Dechert, S., Demeshko, S., Kang, L. Q., Peredkov, S., DeBeer, S., Meyer, F. (2024). Cooperative Sulfur Transformations at a Dinickel Site: A Metal Bridging Sulfur Radical and Its H-Atom Abstraction Thermochemistry. Journal of the American Chemical Society, 146 (33), 23158-23170. doi:10.1021/jacs.4c05113.
Souilah, C., Jannuzzi, S. A. V., Becker, F. J., Demirbas, D., Jenisch, D., Ivlev, S., Xie, X. L., Peredkov, S., Lichtenberg, C., DeBeer, S., Casitas, A. (2024). Synthesis of Iron(IV) Alkynylide Complexes and Their Reactivity to Form 1,3-Diynes. Angewandte Chemie-International Edition. doi:10.1002/anie.202421222.
Salati, M., Dorchies, F., Wang, J. W., Ventosa, M., González-Carrero, S., Bozal-Ginesta, C., Holub, J., Rüdiger, O., DeBeer, S., Gimbert-Suriñach, C., Durrant, J. R., Ertem, M. Z., Gil-Sepulcre, M., Llobet, A. (2024). Covalent Triazine-Based Frameworks with Ru-tda Based Catalyst Anchored via Coordination Bond for Photoinduced Water Oxidation. Small. doi:10.1002/smll.202406375.
Roemelt, C., Peredkov, S., Neese, F., Roemelt, M., DeBeer, S. (2024). Valence-to-core X-ray emission spectroscopy of transition metal tetrahalides: mechanisms governing intensities. Physical Chemistry Chemical Physics, 26 (29), 19960-19975. doi:10.1039/d4cp00967c.
Rice, D. B., Wong, D. N., Weyhermüller, T., Neese, F., DeBeer, S. (2024). The spin-forbidden transition in iron(IV)-oxo catalysts relevant to two-state reactivity. Science Advances, 10 (26). doi:10.1126/sciadv.ado1603.
Reichle, S., Kang, L. Q., Demirbas, D., Weidenthaler, C., Felderhoff, M., DeBeer, S., Schüth, F. (2024). Mechanocatalytic Synthesis of Ammonia: State of the Catalyst During Reaction and Deactivation Pathway. Angewandte Chemie-International Edition, 63 (14). doi:10.1002/anie.202317038.
Rana, A., Peredkov, S., Behrens, M., DeBeer, S. (2024). Probing the Local Environment in Potassium Salts and Potassium-Promoted Catalysts by Potassium Valence-to-Core X-ray Emission Spectroscopy. Inorganic Chemistry, 63 (35), 16217-16223. doi:10.1021/acs.inorgchem.4c02069.
Puja De, G., Singh, K. K., Jannuzzi, S. A. V., Mohanty, A., Velasco, L., Kulbir, Kumar, P., Marco, J. F., Nagarajan, K., Pecharromán, C., Rodríguez-Pascual, P. M., DeBeer, S., Moonshiram, D., Sen Gupta, S., Dasgupta, J. (2024). Trapping an Elusive Fe(IV)-Superoxo Intermediate Inside a Self-Assembled Nanocage in Water at Room Temperature. J. Am. Chem. Soc., 146 (31), 21729-21741. doi:10.1021/jacs.4c05849.
Porte, V., Milunovic, M. N. M., Knof, U., Leischner, T., Danzl, T., Kaiser, D., Gruene, T., Zalibera, M., Jelemenska, I., Bucinsky, L., Jannuzzi, S. A. V., DeBeer, S., Novitchi, G., Maulide, N., Arion, V.B. (2024). Chemical and Redox Noninnocence of Pentane - 2,4- dione Bis(S- methylisothiosemicarbazone) in Cobalt Complexes and Their Application in Wacker-Type Oxidation. JACS Au, 4 (3), 1166-1183. doi:10.1021/jacsau.4c00005.
Peredkov, S., Pereira, N., Grötzsch, D., Hendel, S., Wallacher, D., DeBeer S. (2024). PINK: a tender X-ray beamline for X-ray emission spectroscopy. Journal of Synchrotron Radiation, 31, 622-634. doi:10.1107/s1600577524002200.
Munzone, A., Pujol, M., Tamhankar, A., Joseph, C., Mazurenko, I., Réglier, M., Jannuzzi, S. A. V., Royant, A., Sicoli, G., DeBeer, S., Orio, M., Simaan, A. J., Decroos, C. (2024). Integrated Experimental and Theoretical Investigation of Copper Active Site Properties of a Lytic Polysaccharide Monooxygenase from Serratia marcescens. Inorganic Chemistry, 63 (24), 11063-11078. doi:10.1021/acs.inorgchem.4c00602.
Luo, Z. M., Wang, J. W., Nikolaou, V., Garcia-Padilla, E., Gil-Sepulcre, M., Benet-Buchholz, J., Rüdiger, O., DeBeer, S., Maseras, F., Llobet, A. (2024). Molecular Hybrid Materials for Selective CO2 Electroreduction to Multicarbon Products. Advanced Energy Materials, 14 (43). doi:10.1002/aenm.202402070.
Le, L. N. V., Joyce, J. P., Oyala, P. H., DeBeer, S., Agapie, T. (2024). Highly Activated Terminal Carbon Monoxide Ligand in an Iron-Sulfur Cluster Model of FeMco with Intermediate Local Spin State at Fe. Journal of the American Chemical Society, 146 (8), 5045-5050. doi:10.1021/jacs.3c12025.
Kumar, A., Gil-Sepulcre, M., Lee, J. S., Bui, V. Q., Wang, Y., Rüdiger, O., Kim, M. G., DeBeer, S., Tüysüz, H. (2024). Iridium Single-Atom-Ensembles Stabilized on Mn-Substituted Spinel Oxide for Durable Acidic Water Electrolysis. Advanced Materials, 36 (46). doi:10.1002/adma.202401648.
Kumar, A., Gil-Sepulcre, M., Fandré, J. P., Rüdiger, O., Kim, M. G., DeBeer, S., Tüysüz, H. (2024). Regulating Local Coordination Sphere of Ir Single Atoms at the Atomic Interface for Efficient Oxygen Evolution Reaction. Journal of the American Chemical Society, 146 (48), 32953-32964. doi:10.1021/jacs.4c08847.
Keilwerth, M., Mao, W. Q., Malischewski, M., Jannuzzi, S. A. V., Breitwieser, K., Heinemann, F. W., Scheurer, A., DeBeer, S., Munz, D., Bill, E., Meyer, K. (2024). The synthesis and characterization of an iron(VII) nitrido complex. Nature Chemistry, 16 (4). doi:10.1038/s41557-023-01418-4.
He, B., Hosseini, P., Escalera-López, D., Schulwitz, J., Rüdiger, O., Hagemann, U., Heidelmann, M., DeBeer, S., Muhler, M., Cherevko, S., Tschulik, K., Li, T. (2024). Effects of Dynamic Surface Transformation on the Activity and Stability of Mixed Co-Mn Cubic Spinel Oxides in the Oxygen Evolution Reaction in Alkaline Media. Advanced Energy Materials. doi:10.1002/aenm.202403096.
Hahn, A. W., Zsombor-Pindera, J., Kennepohl, P., DeBeer, S. (2024). Introducing SpectraFit: An Open-Source Tool for Interactive Spectral Analysis. Acs Omega, 9 (22), 23252-23265. doi:10.1021/acsomega.3c09262.
Geoghegan, B. L., Bilyj, J. K., Bernhardt, P. V., DeBeer, S., Cutsail, G. E., III (2024). X-ray absorption and emission spectroscopy of N2S2 Cu(ii)/(iii) complexes. Dalton Transactions, 53 (18), 7828-7838. doi:10.1039/d4dt00085d.
Fataftah, M. S., Wilson, D. W. N., Mathe, Z., Gerard, T. J., Mercado, B. Q., DeBeer, S., Holland, P. L. (2024). Inserting Three-Coordinate Nickel into 4Fe-4S Clusters. ACS Central Science, 10 (10), 1910-1919. doi:10.1021/acscentsci.4c00985.
Debnath, S., Laxmi, S., Stepanic, O. M., Quek, S. Y., van Gastel, M., DeBeer, S., Kramer, T., England, J. (2024). A Four-Coordinate End-On Superoxocopper(II) Complex: Probing the Link between Coordination Number and Reactivity. Journal of the American Chemical Society, 146 (34), 23704-23716. doi:10.1021/jacs.3c12268.
Chen, Z., Fan, Q., Wang, X., Kang, L., Wan, W., Avaro, J. T., Zhang, S., Scheu, C., Zhou, J., DeBeer, S., Heumann, S., Huang, M., Jiang, H., Cölfen, H. (2024). Mechanistic insights into the formation of hydroxides with unconventional coordination environments to achieve their cost-effective synthesis. National Science Review, nwae427. doi: 10.1093/nsr/nwae427.
Chen, S. L., Jelic, J., Rein, D., Najafishirtari, S., Schmidt, F. P., Girgsdies, F., Kang, L. Q., Wandzilak, A., Rabe, A., Doronkin, D. E., Wang, J. H., Ortega, K. F., DeBeer, S., Grunwaldt, J. D., Schlögl, R., Lunkenbein, T., Studt, F., Behrens, M. (2024). Highly loaded bimetallic iron-cobalt catalysts for hydrogen release from ammonia. Nature Communications, 15 (1). doi:10.1038/s41467-023-44661-6.
Castillo, R. G., Van Kuiken, B. E., Weyhermüller, T., DeBeer, S. (2024). Experimentally Assessing the Electronic Structure and Spin-State Energetics in MnFe Dimers Using 1s3p Resonant Inelastic X-ray Scattering. Inorganic Chemistry, 63 (39), 18468-18483. doi:10.1021/acs.inorgchem.4c01538.
2023
Zhang, Y. Y., El Sayed, S., Kang, L. Q., Sanger, M., Wiegand, T., Jessop, P. G., DeBeer, S., Bordet, A., Leitner, W. (2023). Adaptive Catalysts for the Selective Hydrogenation of Bicyclic Heteroaromatics using Ruthenium Nanoparticles on a CO2-Responsive Support. Angewandte Chemie-International Edition, 62 (48). doi:10.1002/anie.202311427.
Yogendra, S., Wilson, D. W. N., Hahn, A. W., Weyhermüller, T., Van Stappen, C., Holland, P., DeBeer, S. (2023). Sulfur-Ligated 2Fe-2C Clusters as Synthetic Model Systems for Nitrogenase. Inorganic Chemistry, 62 (6), 2663-2671. doi:10.1021/acs.inorgchem.2c03693.
Wandzilak, A., Grubel, K., Skubi, K. L., McWilliams, S. F., Bessas, D., Rana, A., Hugenbruch, S., Dey, A., Holland, P. L., DeBeer, S. (2023). Mossbauer and Nuclear Resonance Vibrational Spectroscopy Studies of Iron Species Involved in N-N Bond Cleavage. Inorganic Chemistry, 62 (45), 18449-18464. doi:10.1021/acs.inorgchem.3c02594.
Velasco, L., Liu, C. M., Zhang, X. Y., Grau, S., Gil-Sepulcre, M., Gimbert-Suriñach, C., Picón, A., Llobet, A., DeBeer, S., Moonshiram, D. (2023). Mapping the Ultrafast Mechanistic Pathways of Co Photocatalysts in Pure Water through Time-Resolved X-ray Spectroscopy. ChemSusChem, 16 (21), e202300719. doi:10.1002/cssc.202300719.
Van Stappen, C., Benediktsson, B., Rana, A., Chumakov, A., Yoda, Y., Bessas, D., Decamps, L., Bjornsson, R., DeBeer, S. (2023). Structural correlations of nitrogenase active sites using nuclear resonance vibrational spectroscopy and QM/MM calculations. Faraday Discussions, 243 (0), 253-269. doi:10.1039/d2fd00174h.
Sodreau, A., Zahedi, H. G., Dervisoglu, R., Kang, L. Q., Menten, J., Zenner, J., Terefenko, N., DeBeer, S., Wiegand, T., Bordet, A., Leitner, W. (2023). A Simple and Versatile Approach for the Low-Temperature Synthesis of Transition Metal Phosphide Nanoparticles from Metal Chloride Complexes and P(SiMe3)3. Advanced Materials, 35 (49). doi:10.1002/adma.202306621.
Pielsticker, L., Nicholls, R. L., DeBeer, S., Greiner, M. (2023). Convolutional neural network framework for the automated analysis of transition metal X-ray photoelectron spectra. Analytica Chimica Acta, 1271. doi:10.1016/j.aca.2023.341433.
Pang, Y., Nöthling, N., Leutzsch, M., Kang, L. Q., Bill, E., van Gastel, M., Reijerse, E., Goddard, R., Wagner, L., SantaLucia, D., DeBeer, S., Neese, F., Cornella, J. (2023). Synthesis and isolation of a triplet bismuthinidene with a quenched magnetic response. Science, 380 (6649), 1043-1048. doi:10.1126/science.adg2833.
Martini, M. A., Bikbaev, K., Pang, Y. J., Lorent, C., Wiemann, C., Breuer, N., Zebger, I., DeBeer, S., Span, I., Bjornsson, R., Birrell, J. A., Rodríguez-Maciá, P. (2023). Binding of exogenous cyanide reveals new active-site states in FeFe hydrogenases. Chemical Science, 14 (11), 2826-2838. doi:10.1039/d2sc06098a.
Mao, W. Q., Zhang, Z. H., Fehn, D., Jannuzzi, S. A. V., Heinemann, F. W., Scheurer, A., van Gastel, M., DeBeer, S., Munz, D., Meyer, K. (2023). Synthesis and Reactivity of a Cobalt-Supported Singlet Nitrene. Journal of the American Chemical Society, 145 (25), 13650-13662. doi:10.1021/jacs.3c01478.
Liu, Y., Chatterjee, S., Cutsail, G. E., III, Peredkov, S., Gupta, S. K., Dechert, S., DeBeer, S., Meyer, F. (2023). Cu4S Cluster in "0-Hole" and "1-Hole" States: Geometric and Electronic Structure Variations for the Active CuZ* Site of N2O Reductase. Journal of the American Chemical Society. doi:10.1021/jacs.3c04893.
Kovel, C. B., Darmon, J. M., Stieber, S. C. E., Pombar, G., Pabst, T. P., Theis, B., Turner, Z. R., Üngör, Ö., Shatruk, M., DeBeer, S., Chirik, P. J. (2023). Bimolecular Reductive Elimination of Ethane from Pyridine(diimine) Iron Methyl Complexes: Mechanism, Electronic Structure, and Entry into 2+2 Cycloaddition Catalysis. Journal of the American Chemical Society. doi:10.1021/jacs.2c10547.
Keilwerth, M., Mao, W. Q., Jannuzzi, S. A. V., Grunwald, L., Heinemann, F. W., Scheurer, A., Sutter, J., DeBeer, S., Munz, D., Meyer, K. (2023). From Divalent to Pentavalent Iron Imido Complexes and an Fe(V) Nitride via N-C Bond Cleavage. Journal of the American Chemical Society, 145 (2), 873-887. doi:10.1021/jacs.2c09072.
Hou, K. P., Börgel, J., Jiang, H. Z. H., SantaLucia, D. J., Kwon, H., Zhuang, H., Chakarawet, K., Rohde, R. C., Taylor, J. W., Dun, C. C., Paley, M. V., Turkiewicz, A. B., Park, J. G., Mao, H. Y., Zhu, Z. T., Alp, E. E., Zhao, J. Y., Hu, M. Y., Lavina, B., Peredkov, S., Lv, X. D., Oktawiec, J., Meihaus, K. R., Pantazis, D. A., Vandone, M., Colombo, V., Bill, E., Urban, J. J., Britt, R. D., Grandjean, F., Long, G. J., DeBeer, S., Neese, F., Reimer, J. A., Long, J. R. (2023). Reactive high-spin iron(IV)-oxo sites through dioxygen activation in a metal-organic framework. Science, 382 (6670), 547-553. doi:10.1126/science.add7417.
Hau, J. L., Kaltwasser, S., Muras, V., Casutt, M. S., Vohl, G., Claussen, B., Steffen, W., Leitner, A., Bill, E., Cutsail, G. E., DeBeer, S., Vonck, J., Steuber, J., Fritz, G. (2023). Conformational coupling of redox-driven Na+-translocation in Vibrio cholerae NADH:quinone oxidoreductase. Nature Structural & Molecular Biology, 30 (11). doi:10.1038/s41594-023-01099-0.
Hall, K. R., Joseph, C., Ayuso-Fernández, I., Tamhankar, A., Rieder, L., Skaali, R., Golten, O., Neese, F., Rohr, Å., Jannuzzi, S. A. V., DeBeer, S., Eijsink, V. G. H., Sorlie, M. (2023). A Conserved Second Sphere Residue Tunes Copper Site Reactivity in Lytic Polysaccharide Monooxygenases. Journal of the American Chemical Society. doi:10.1021/jacs.3c05342.
Ghaida, F. A., Brinkert, K., Chen, P., DeBeer, S., Hoffman, B. M., Holland, P. L., Laxmi, S., MacFarlane, D., Peters, J. C., Peters, J. W., Pickett, C. J., Seefeldt, L. C., Shylin, S. I., Stephens, I. E. L., Vincent, K. A., Wang, Q. R., Westhead, O. (2023). Enzymatic N2 activation: general discussion. Faraday Discussions, 243, 287-295. doi:10.1039/d3fd90011h.
Genoux, A., Pauly, M., Rooney, C. L., Choi, C., Shang, B., McGuigan, S., Fataftah, M. S., Kayser, Y., Suhr, S. C. B., DeBeer, S., Wang, H. L., Maggard, P. A., Holland, P. L. (2023). Well-Defined Iron Sites in Crystalline Carbon Nitride. Journal of the American Chemical Society, 145 (38), 20739-20744. doi:10.1021/jacs.3c05417.
Erbe, A., Tesch, M. F., Ruediger, O., Kaiser, B., DeBeer, S., Rabe, M. (2023). Operando studies of Mn oxide based electrocatalysts for the oxygen evolution reaction. Physical Chemistry Chemical Physics, 25 (40), 26958-26971. doi:10.1039/d3cp02384b.
Chrysina, M., Drosou, M., Castillo, R. G., Reus, M., Neese, F., Krewald, V., Pantazis, D. A., DeBeer, S. (2023). Nature of S-States in the Oxygen-Evolving Complex Resolved by High-Energy Resolution Fluorescence Detected X-ray Absorption Spectroscopy. Journal of the American Chemical Society, 145 (47), 25579-25594. doi:10.1021/jacs.3c06046.
Caserta, G., Hartmann, S., Van Stappen, C., Karafoulidi-Retsou, C., Lorent, C., Yelin, S., Keck, M., Schoknecht, J., Sergueev, I., Yoda, Y., Hildebrandt, P., Limberg, C., DeBeer, S., Zebger, I., Frielingsdorf, S., Lenz, O. (2023). Stepwise assembly of the active site of NiFe-hydrogenase. Nature Chemical Biology, 19 (4), 498. doi:10.1038/s41589-022-01226-w.
Anandaraj, S. J. L., Kang, L. Q., DeBeer, S., Bordet, A., Leitner, W. (2023). Catalytic Hydrogenation of CO2 to Formate Using Ruthenium Nanoparticles Immobilized on Supported Ionic Liquid Phases. Small, 19 (18). doi:10.1002/smll.202206806.
2022
Yu, M. Q., Weidenthaler, C., Wang, Y., Budiyanto, E., Sahin, E. O., Chen, M. M., DeBeer, S., Rüdiger, O., Tüysüz, H. (2022). Surface Boron Modulation on Cobalt Oxide Nanocrystals for Electrochemical Oxygen Evolution Reaction. Angewandte Chemie-International Edition, 61 (42). doi:10.1002/anie.202211543.
Xiang, W. K., Yang, N. T., Li, X. P., Linnemann, J., Hagemann, U., Ruediger, O., Heidelmann, M., Falk, T., Aramini, M., DeBeer, S., Muhler, M., Tschulik, K., Li, T. (2022). 3D atomic-scale imaging of mixed Co-Fe spinel oxide nanoparticles during oxygen evolution reaction. Nature Communications, 13 (1). doi:10.1038/s41467-021-27788-2.
Van Stappen, C., Jiménez-Vicente, E., Pérez-González, A., Yang, Z. Y., Seefeldt, L. C., DeBeer, S., Dean, D. R., Decamps, L. (2022). A conformational role for NifW in the maturation of molybdenum nitrogenase P-cluster. Chemical Science, 13 (12), 3489-3500. doi:10.1039/d1sc06418e.
Souilah, C., Jannuzzi, S. A. V., Demirbas, D., Ivlev, S., Swart, M., DeBeer, S., Casitas, A. (2022). Synthesis of FeIII and FeIV Cyanide Complexes Using Hypervalent Iodine Reagents as Cyano-Transfer One-Electron Oxidants. Angewandte Chemie-International Edition, 61 (22), e202201699. doi:10.1002/anie.202201699.
Sisodiya-Amrute, S., Van Stappen, C., Rengshausen, S., Han, C. H., Sodreau, A., Weidenthaler, C., Tricard, S., DeBeer, S., Chaudret, B., Bordet, A., Leitner, W. (2022). Bimetallic MxRu100-x nanoparticles (M = Fe, Co) on supported ionic liquid phases (MxRu100-x @SILP) as hydrogenation catalysts: Influence of M and M: Ru ratio on activity and selectivity. Journal of Catalysis, 407, 141-148. doi:10.1016/j.jcat.2022.01.030.
Mao, W. Q., Fehn, D., Heinemann, F. W., Scheurer, A., van Gastel, M., Jannuzzi, S. A., DeBeer, S., Munz, D., Meyer, K. (2022). Umpolung in a Pair of Cobalt(III) Terminal Imido/Imidyl Complexes. Angewandte Chemie-International Edition, 61 (36). doi:10.1002/anie.202206848.
Levin, N., Casadevall, C., Cutsail, G. E., Lloret-Fillol, J., DeBeer, S., Rüdiger, O. (2022). XAS and EPR in Situ Observation of Ru(V) Oxo Intermediate in a Ru Water Oxidation Complex**. Chemelectrochem, 9 (3). doi:10.1002/celc.202101271.
Jedrzkiewicz, D., Mai, J., Langer, J., Mathe, Z., Patel, N., DeBeer, S., Harder, S. (2022). Access to a Labile Monomeric Magnesium Radical by Ball-Milling. Angewandte Chemie-International Edition, 61 (15). doi:10.1002/anie.202200511.
Henthorn, J. T., DeBeer, S. (2022). Selenium Valence-to-Core X-ray Emission Spectroscopy and Kβ HERFD X-ray Absorption Spectroscopy as Complementary Probes of Chemical and Electronic Structure. Inorganic Chemistry, 61 (6), 2760-2767. doi:10.1021/acs.inorgchem.1c02802.
Henthorn, J. T., Cutsail, G. E., Weyhermüller, T., DeBeer, S. (2022). Stabilization of intermediate spin states in mixed-valent diiron dichalcogenide complexes. Nature Chemistry, 14 (3), 328. doi:10.1038/s41557-021-00853-5.
Geoghegan, B. L., Liu, Y., Peredkov, S., Dechert, S., Meyer, F., DeBeer, S., Cutsail, G. E. (2022). Combining Valence-to-Core X-ray Emission and Cu K-edge X-ray Absorption Spectroscopies to Experimentally Assess Oxidation State in Organometallic Cu(I)/(II)/(III) Complexes. Journal of the American Chemical Society, 144 (6), 2520-2534. doi:10.1021/jacs.1c09505.
Decamps, L., Rice, D. B., DeBeer, S. (2022). An Fe6C Core in All Nitrogenase Cofactors. Angewandte Chemie-International Edition, 61 (41). doi:10.1002/anie.202209190.
Czastka, K., Oughli, A. A., Rüdiger, O., DeBeer, S. (2022). Enzymatic X-ray absorption spectroelectrochemistry. Faraday Discussions, 234, 214-231. doi:10.1039/d1fd00079a.
Cutsail, G. E., DeBeer, S. (2022). Challenges and Opportunities for Applications of Advanced X-ray Spectroscopy in Catalysis Research. Acs Catalysis, 12 (10), 5864-5886. doi:10.1021/acscatal.2c01016.
Cutsail, G. E., III, Banerjee, R., Rice, D. B., Stepanic, O. M., Lipscomb, J. D., DeBeer, S. (2022). Determination of the iron(IV) local spin states of the Q intermediate of soluble methane monooxygenase by Kβ X-ray emission spectroscopy. Journal of Biological Inorganic Chemistry, 27 (6), 573-582. doi:10.1007/s00775-022-01953-4.
Cutsail, G., III, Schott-Verdugo, S., Müller, L., DeBeer, S., Groth, G., Gohlke, H. (2022). Spectroscopic and QM/MM studies of the Cu(I) binding site of the plant ethylene receptor ETR1. Biophysical Journal, 121 (20), 3862-3873. doi:10.1016/j.bpj.2022.09.007.
Chatterjee, S., Harden, I., Bistoni, G., Castillo, R. G., Chabbra, S., van Gastel, M., Schnegg, A., Bill, E., Birrell, J. A., Morandi, B., Neese, F., DeBeer, S. (2022). A Combined Spectroscopic and Computational Study on the Mechanism of Iron-Catalyzed Aminofunctionalization of Olefins Using Hydroxylamine Derived N-O Reagent as the "Amino" Source and "Oxidant". Journal of the American Chemical Society, 144 (6), 2637-2656. doi:10.1021/jacs.1c11083.
Bowker, M., DeBeer, S., Dummer, N. F., Hutchings, G. J., Scheffler, M., Schüth, F., Taylor, S. H., Tüysüz, H. (2022). Advancing Critical Chemical Processes for a Sustainable Future: Challenges for Industry and the Max Planck-Cardiff Centre on the Fundamentals of Heterogeneous Catalysis (FUNCAT). Angewandte Chemie-International Edition, 61 (50), e202209016. doi:10.1002/anie.202209016.
Anilkumar, A., Ash, P., Chakravarty, A. R., Comba, P., DeBeer, S., Dey, A., Draksharapu, A., Goswami, D., Itoh, S., Karlin, K., Lakshmi, K. V., Mazumdar, S., Pantazis, D., Parker, D., Que, L. C., Rajaraman, G., Rath, S. P., Sastri, C., Sen Gupta, S., Solomon, E. I. (2022). Electron transfer, spectroscopy and theory: general discussion. Faraday Discussions, 234 (0), 245-263. doi:10.1039/d2fd90013k.
Alkan, B., Braun, M., Landrot, G., Rüdiger, O., Andronescu, C., DeBeer, S., Schulz, C., Wiggers, H. (2022). Spray-flame-synthesized Sr- and Fe-substituted LaCoO3 perovskite nanoparticles with enhanced OER activities. Journal of Materials Science, 57 (40), 18923-18936. doi:10.1007/s10853-022-07738-z.
Aldous, L., Comba, P., DeBeer, S., Dey, A., Draksharapu, A., Duboc, C., Itoh, S., Karlin, K., Kundu, S., Domene, R. L., Maréchal, J. D., Mazumdar, S., Mukherjee, R., Parker, D., Pordea, A., Que, L. C., Rath, S. P., Sadler, P., Sastri, C., Schindler, S., Schünemann, V., Sen Gupta, S., Solomon, E. I., Stack, T. D. P. (2022). Small molecule activation and synthetic analogues: general discussion. Faraday Discussions, 234 (0), 129-142. doi:10.1039/d2fd90012b.
Awards
- 2024 Michael Lappert Lectureship Award
- 2023 Glenn T. Seaborg Lectureship, University of California Berkeley
- 2022 R.J.P. Williams Lectureship, Oxford University
- 2022 Malcom H. Chisholm Lecturer, The Ohio State University
- 2021 Fellow of the Royal Society of Chemistry
- 2019 European Research Council Synergy Grant Awardee
- 2016 Inorganic Chemistry Lectureship Award
- 2015 Society of Biological Inorganic Chemistry, Early Career Award
- 2013 European Research Council Consolidator Grant Awardee
- 2012 Kavli Fellow, U.S. National Academy of Sciences
- 2011-2013 Alfred P. Sloan Research Fellow
Group members
Office
Valerie EstersGroup leaders
Dr. Rebeca Gomez CastilloDr. Sergey Peredkov
Dr. Christina Römelt
Dr. Olaf Rüdiger
Dr. Kushal Sengupta
Dr. Thomas Weyhermüller
Project leaders
Dr. Yves KayserDr. Sergio Augusto Venturinelli Jannuzzi
Inorganic Spectroscopy
Our department focuses on the development and application of advanced X-ray spectroscopic tools for understanding processes in biological and chemical catalysis. We are interested in the development of both static and time-dependent probes of transition metal electronic structure, and in particular the development of two-dimensional X-ray spectroscopic probes, which enable enhanced selectivity. Challenging questions in energy research motivate our research. Namely how can earth-abundant base metals enable the activation of strong chemical bonds? In this context, we focus on four transition-metal catalyzed reactions:
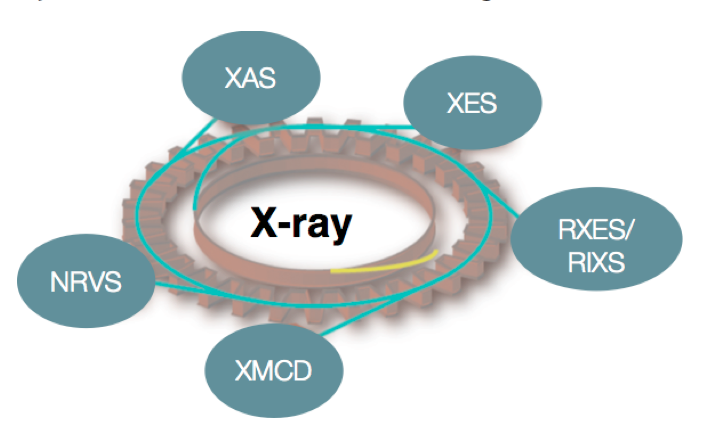
In all of the reactions shown above, there are both biological (homogenous) and heterogeneous catalysts, which are able to effect these conversions, albeit with varied efficiencies and in some cases only in multiple steps (e.g. industrial methane oxidation). We are interested in understanding the electronic structure of the catalysts and the transformations, which occur over the course of a reaction. Ideally this knowledge will form a fundamental basis for rational catalytic design. Our approach utilizes X-ray based spectroscopies, including resonant (RXES, RIXS) and non-resonant X-ray emission (XES), X-ray absorption (XAS), X-ray magnetic circular dichroism (XMCD), and nuclear resonant vibrational spectroscopy (NRVS) (Figure 1). In addition to our synchrotron-based studies, we are developing instrumentation for in-house X-ray spectroscopy. The applied combination of spectroscopic approaches provides insight into metal oxidation states, spin states, and the local coordination environments of the transition metal active sites. Our spectroscopic studies are coupled to more standard laboratory spectroscopy (UV-Vis, EPR, Raman and Mössbauer) and modern computational methods, in order to obtain quantitative insights into electronic structural changes in catalytic systems.

1. Spectroscopy Developments
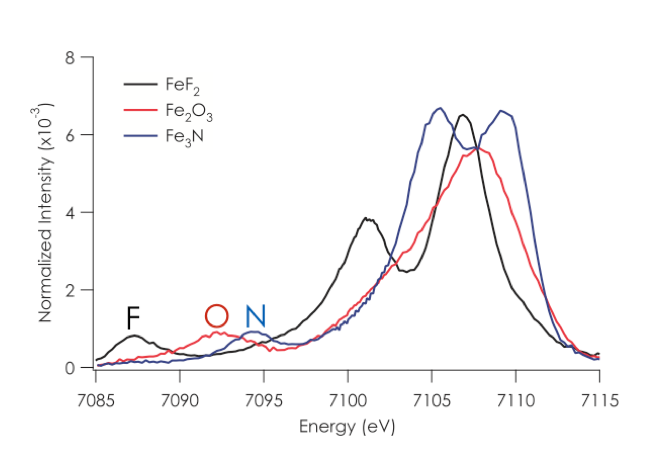
1.1. Valence XES. Recent work in our research group has focused on developing the full information content of valence XES and applying this method to biological catalysts.1-12 In a valence XES measurement, one first ionizes a metal 1s electron and then monitors the resultant fluorescence after a valence electron refills the metal 1s core hole. As such, valence XES spectra provide a map of the ligand ionization potentials, and show a strong dependence on both ligand identity (i.e. C, N, or O) and protonation state (O2-, OH-, or H2O) (Figure 2). A notable application of this method was the use of valence XES to identify central carbon atom in the FeMoco cluster of nitrogenase.8 As the identification of the central atom had eluded characterization by other experimental approaches for almost a decade, this result serves to highlight the power and selectivity of the method. More recent studies in my group have focused on the use of valence XES to understand metallocofactor cluster biosynthesis and the atomic composition of FeVco in Vanadium nitrogenase.7, 13, 14
Valence XES is sensitive not only to the identity of ligand, but also to the degree of activation (i.e. weakening) of a bound ligand. For instance, when performing valence XES on a complex with an Fe-N2 moiety, spectral features which can be correlated with the N2 2s2s σ* orbital interacting with the Fe centers have been identified.12 As the N-N bond is lengthened, the energy of this transition decreases in energy, with a shift of more than 2 eV on going from an N-N triple bond to a cleaved nitride. This means that Fe valence XES is an ideal spectral probe of N-N bond cleavage (or cleavage of other diatomic ligands) by Fe (or any other transition metal).
To this end, we are developing dispersive valence XES, as a time-resolved probe of transition metal catalysis. Recently, funded by an ERC Consolidator Grant (N2RED, PI: DeBeer) and in collaboration with the group of Prof. Kangießer at TU Berlin, an in-house dispersive XES setup has been designed and commissioned. The instrument utilizes a laboratory X-ray source (Excillum, Metal Jet) in combination with a cylindrical Highly Annealed Pyrolytic Graphite (HAPG) crystal and a CCD detector to obtain XES in the 2.4-9 keV range (Figure 3). This unique in-house setup helps ease demands on synchrotron time, providing an ideal setup for concentrated samples and a means to obtain preliminary measurements before conducting more complex synchrotron or X-ray free electron laser measurements. In the context of synchrotron-based dispersive XES, the Department of Inorganic Spectroscopy is also leading development of the PINK beam line at the Energy Materials In-situ Laboratory in Berlin. PINK will provide an intense source for simultaneous “two-color” XES measurements of heterometallic catalysts. These efforts are lead by group leader Dr. Sergey Peredkov.
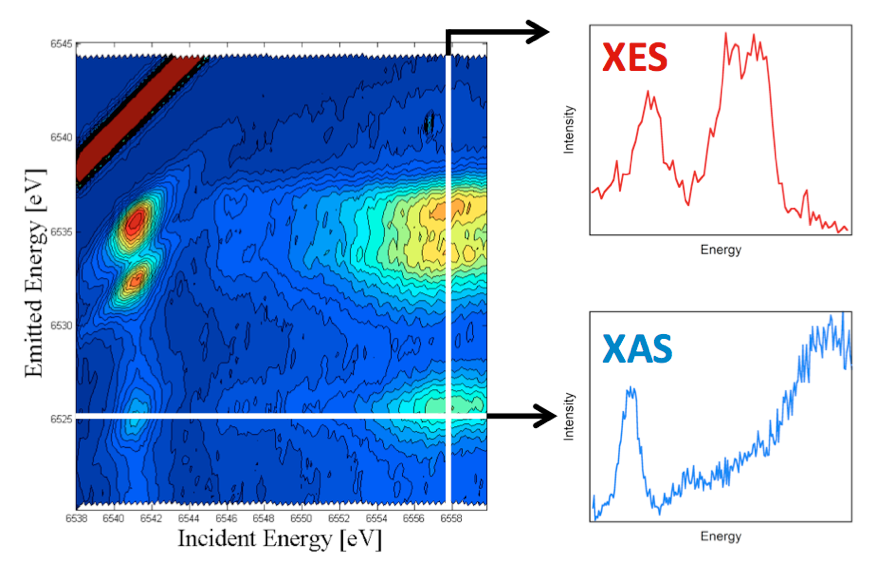
1.2 Resonant valence XES. To further enhance the selectivity of valence XES, we are developing resonant valence XES.2 This is a two-dimensional measurement where one resonantly excites a metal 1s core electron into the partially occupied and unoccupied states localized on the photoabsorber (i.e. the equivalent of an X-ray absorption edge scan), while simultaneously monitoring the highest energy emission processes (i.e. the valence XES). In doing so, one is able to construct a two-dimensional plane, with the incident energy on the x-axis and the emission on the y-axis (Figure 4). The horizontal cuts through this plane, then represent the XAS data at a given emission energy and effectively correspond to “ligand selective XAS”. While the vertical cuts represent the XES data at a given incident energy or valence RXES. We believe this approach should be of particular utility for detecting the XAS of different types of metal-ligand interactions within complex media.
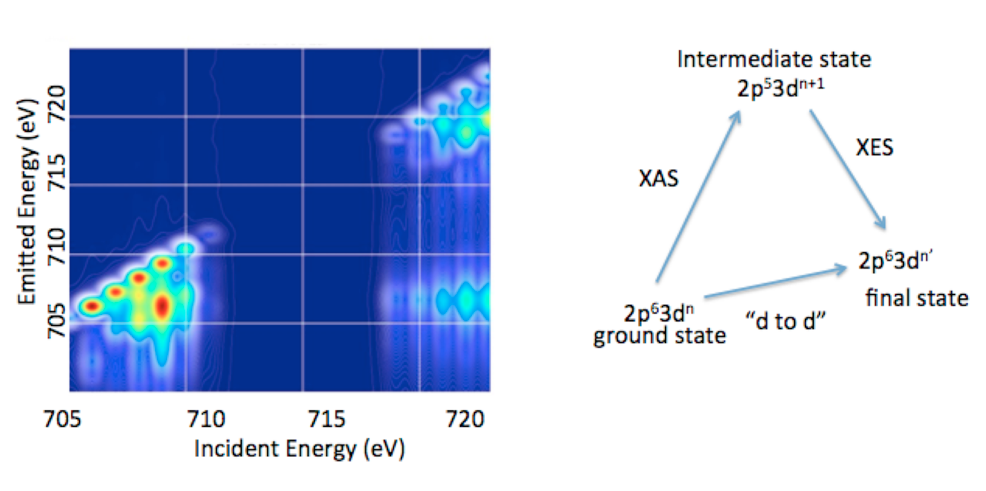
1.3. 2p3d RIXS. In addition to valence RXES, we are developing the full information content of transition metal 2p3d RXES or what is more commonly referred to as 2p3d RIXS.15 2p3d RIXS involves the dipole allowed excitation of a 2p63dn ground state to arrive at a2p53dn+1 intermediate state, which subsequently decays via a dipole allowed process to a 2p63dn’final state (Figure 5). Hence the difference between the ground state and the final state corresponds to formal dipole forbidden d-to-d transitions, which one arrives at through two dipole allowed processes. In a 2p3d RIXS measurement, the energy difference between the ground state and the final state is referred to as the “energy transfer” axis, which due to the nature of the experiment may be readily measured from 0-10 eV (or from 0 to over 80,000 cm-1). This means that spectral regions that cannot be accessed using standard optical or infrared spectroscopic probes are accessible. In addition, 2p3d RIXS experiments have been shown, in certain cases, to allow for the observation of formally spin forbidden transitions due to intermediate state spin orbit coupling. Hence, it may be possible to effectively measure the complete spin ladder, thus providing unprecedented experimental insights into transition metal electronic structure.
2. Applications
The spectroscopy developments discussed in the proceeding sections form a strong basis for applications in biological and chemical catalysis. In the section that follows, we briefly highlight some of our ongoing and planned efforts.
N2 reduction. The transformation of N2 to ammonia (NH3) is crucial for life on earth. This kinetically challenging processes is enabled biologically by the enzyme nitrogenase and industrially by the Haber-Bosch process, with remarkable efficiencies. In contrast, chemists have yet to synthesize a molecular system that can compete with either the biological or the industrial process. It is our view that inability to design effective molecular catalysts derives in part from a lack of fundamental knowledge. How does one effectively cleave N2, the strongest homodiatomic bond in known in chemistry? How does the metal catalyst interact with N2? What intermediate species are generated in the process of N2 bond breaking? To answer these questions we are applying advanced x-ray spectroscopic methods to both the biological and the heterogeneous catalysts. For the biological catalyst, we seek to understand the complex electronic structure of the ground state, the role of the central carbon, the role of the heterometal (Mo or V) and the site of substrate interaction. For the heterogeneous catalysts, we seek to understand the nature of the surface nitrides, the oxidation state of the “active” Fe and the interaction of the potassium promoter. These questions will be answered using the advanced spectroscopic approaches detailed in the preceding sections.
CH4 Oxidation. The biological oxidation of methane to methanol by soluble methane monoxygenase is a process of fundamental interest in the context of a sustainable energy economy. Presently we are focusing on elucidating the mechanism of biological methane oxidation, and in particular determining the controversial structure of the intermediate MMO-Q, which is the species responsible for extracting a proton from inert methane. Utilizing high-resolution XAS methods, we have recently obtained new insights into the elextronic structure of MMO-Q. This motivates further freeze-quench, as well as in situ studies, of biological methane oxidation.
H2O Oxidation. Similar to our studies of N2 reduction, we are also interested in understanding the process of Mn-mediated water oxidation at both the biological and heterogeneous limits. To this end, we utilize advanced X-ray spectroscopic methods to examine Mn birnessites, surface doped Mn systems, and the Mn4Ca active site of photosystem II. “Two-color” simultaneous Mn and Ca XES will be utilized in a time-resolved fashion to evaluate the role of both the Mn and the Ca in catalysis. Our goal is to obtain atomic level insights into the factors that govern O-O bond formation.
H2 production. Hydrogen production and activation has been the focus of extensive research over the last few decades in the context of renewable energy and carbon-neutral fuels. The most prevalent and robust hydrogenases contain a heterobimetallic [NiFe] active site. The redox-active nickel center is thought to play a critical role in hydrogen binding and activation, and many prior studies using x-ray crystallography, electrochemistry, FTIR, and EPR spectroscopy have been carried out to characterize the activation and mechanism of the enzyme. However, of the three catalytically relevant states, only the Ni-C state is paramagnetic. Thus, along with the aerobically-inactivated, hydroxide-bridged Ni-B state, the hydride-bridged Ni-C state represents the only catalytic intermediate that has been structurally well-characterized. Many approaches have been attempted to characterize the other two intermediates, the Ni-SI and Ni-R states, including FTIR and X-ray absorption spectroscopy. However, a number of questions remain, including identification of the nickel spin states, nature of the bridging ligand between the two metal centers in each redox state, metal to which dihydrogen coordinates, and mode of hydrogen binding to that metal. These questions will be addressed through a combination of advanced X-ray based approaches and should provide fundamental insights into the mechanism of biological H2 production.
References
[1] Delgado-Jaime, M. U., Dible, B. R., Chiang, K. P., Brennessel, W. W., Bergmann, U., Holland, P. L., and DeBeer, S. (2011) Identification of a Single Light Atom within a Multinuclear Metal Cluster Using Valence-to-Core X-ray Emission Spectroscopy, Inorg Chem 50, 10709-10717.
[2] Hall, E. R., Pollock, C. J., Bendix, J., Collins, T. J., Glatzel, P., and DeBeer, S. (2014) Valence-to-Core-Detected X-ray Absorption Spectroscopy: Targeting Ligand Selectivity, J Am Chem Soc 136, 10076-10084.
[3] Hugenbruch, S., Shafaat, H. S., Kramer, T., Delgado-Jaime, M. U., Weber, K., Neese, F., Lubitz, W., and DeBeer, S. (2016) In search of metal hydrides: an X-ray absorption and emission study of [NiFe] hydrogenase model complexes, Phys Chem Chem Phys 18, 10688-10699.
[4] Kowalska, J., and DeBeer, S. (2015) The role of X-ray spectroscopy in understanding the geometric and electronic structure of nitrogenase, Bba-Mol Cell Res 1853, 1406-1415.
[5] Kowalska, J. K., Hahn, A. W., Albers, A., Schiewer, C. E., Bjornsson, R., Lima, F. A., Meyer, F., and DeBeer, S. (2016) X-ray Absorption and Emission Spectroscopic Studies of [L2Fe2S2](n) Model Complexes: Implications for the Experimental Evaluation of Redox States in Iron-Sulfur Clusters, Inorg Chem 55, 4485-4497.
[6] Kowalska, J. K., Lima, F. A., Pollock, C. J., Rees, J. A., and DeBeer, S. (2016) A Practical Guide to High-resolution X-ray Spectroscopic Measurements and their Applications in Bioinorganic Chemistry, Isr J Chem 56, 803-815.
[7] Lancaster, K. M., Hu, Y. L., Bergmann, U., Ribbe, M. W., and DeBeer, S. (2013) X-ray Spectroscopic Observation of an Interstitial Carbide in NifEN-Bound FeMoco Precursor, J Am Chem Soc 135, 610-612.
[8] Lancaster, K. M., Roemelt, M., Ettenhuber, P., Hu, Y. L., Ribbe, M. W., Neese, F., Bergmann, U., and DeBeer, S. (2011) X-ray Emission Spectroscopy Evidences a Central Carbon in the Nitrogenase Iron-Molybdenum Cofactor, Science 334, 974-977.
[9] Lee, N., Petrenko, T., Bergmann, U., Neese, F., and DeBeer, S. (2010) Probing Valence Orbital Composition with Iron K beta X-ray Emission Spectroscopy, J Am Chem Soc 132, 9715-9727.
[10] Pollock, C. J., and DeBeer, S. (2011) Valence-to-Core X-ray Emission Spectroscopy: A Sensitive Probe of the Nature of a Bound Ligand, J Am Chem Soc 133, 5594-5601.
[11] Pollock, C. J., and DeBeer, S. (2015) Insights into the Geometric and Electronic Structure of Transition Metal Centers from Valence-to-Core X-ray Emission Spectroscopy, Accounts Chem Res 48, 2967-2975.
[12] Pollock, C. J., Grubel, K., Holland, P. L., and DeBeer, S. (2013) Experimentally Quantifying Small-Molecule Bond Activation Using Valence-to-Core X-ray Emission Spectroscopy, J Am Chem Soc 135, 11803-11808.
[13] Rees, J. A., Martin-Diaconescu, V., Kovacs, J. A., and DeBeer, S. (2015) X-ray Absorption and Emission Study of Dioxygen Activation by a Small-Molecule Manganese Complex, Inorg Chem 54, 6410-6422.
[14] Rees, J. A., Bjornsson, R., Kowalska, J. K., Lima, F. A., Schlesier, J., Sippel, D., Weyhermueller, T., Einsle, O., Kovacs, J. A., and DeBeer, S. (2017) Comparative electronic structures of nitrogenase FeMoco and FeVco, Dalton T 46, 2445-2455.
[15] Van Kuiken, B. E., Hahn, A. W., Maganas, D., and DeBeer, S. (2016) Measuring Spin-Allowed and Spin-Forbidden d-d Excitations in Vanadium Complexes with 2p3d Resonant Inelastic X-ray Scattering, Inorg Chem 55, 11497-11501.