Prof. Dr. Walter Leitner - Molekulare Katalyse
- Prof. Dr. Walter Leitner
- Geschäftsführender Direktor
- Molekulare Katalyse
- +49 (0)208 306 - 3614
- walter.leitner(at)cec.mpg.de
- Raum: 611
Vita
| Dipl.-Chem. Univ. (Chemie) | Universität Regensburg (1982-1987) |
| Dr. rer. nat. | Institut für Anorganische Chemie, Universität Regensburg (1987-1989) |
| Postdoc | Dyson Perrins Laboratory for Organic Chemistry, University of Oxford, UK (1990) |
| Stipendium | Liebig-Stipendiat des Fonds der Chemischen Industrie, Universität Regensburg (1991-1992) |
| Wiss. MA | Max-Planck-Arbeitsgruppe CO2-Chemie, Friedrich-Schiller-Universität Jena (1992-1995) |
| Habilitation | Friedrich-Schiller-Universität Jena (1995) |
| Privatdozent | Friedrich-Schiller-Universität Jena (1995) |
| Gruppenleiter | Abteilung 'Organische Synthese', Max-Planck-Institut für Kohlenforschung, Mülheim/Ruhr (1995-1998) |
| Leiter des Technikums | Max-Planck-Institut für Kohlenforschung, Mülheim/Ruhr (1998-2002) |
| Lehrstuhlvertretung | Technische Chemie und Petrolchemie, RWTH Aachen (2000-2002) |
| Lehrstuhlinhaber | Technische Chemie und Petrolchemie, Institut für Technische und Makromolekulare Chemie, RWTH Aachen (seit 2002) |
| Ext. Wiss. Mitglied | Max-Planck-Institut für Kohlenforschung, Mülheim/Ruhr (2002-2017) |
| Direktor | Molekulare Katalyse, MPI CEC (seit 2017) |
Publications
Full publications list | ORCID | ResearcherID
Selected MPI CEC publications
2024
- Zhang, Y. Y., Levin, N., Kang, L. Q., Müller, F., Zobel, M., DeBeer, S., Leitner, W., Bordet, A. (2024). Design and Understanding of Adaptive Hydrogenation Catalysts Triggered by the H2/CO2-Formic Acid Equilibrium. Journal of the American Chemical Society, 146 (44), 30057-30067. doi:10.1021/jacs.4c06765.
- Zenner, J., Tran, K., Kang, L. Q., Kinzel, N. W., Werlé, C., DeBeer, S., Bordet, A., Leitner, W. (2024). Synthesis, Characterization, and Catalytic Application of Colloidal and Supported Manganese Nanoparticles. Chemistry-a European Journal, 30 (25). doi:10.1002/chem.202304228.
- Wessel, N., Medhekar, R. S., Sonnenberg, M., Stieber, H., Leitner, W., Vorholt, A. J. (2024). Catalyst in Sight: The Use of Benchtop NMR Spectrometers to Maintain the Activity of Pd-PPh3 Catalysts. ACS Catalysis, 14 (14), 10679-10688. doi:10.1021/acscatal.4c02606.
- Vossen, J. T., Patzina, F., Leitner, W., Vorholt, A. J. (2024). Studying the Recycling and Deactivation of Rh/Biphephos Complexes in the Isomerization-Hydroformylation Tandem Reaction. Acs Sustainable Chemistry & Engineering. doi:10.1021/acssuschemeng.4c03970.
- Voelker, S., Groll, N., Bachmann, M. Mueller, L., Neumann, M., Kossioris, T., Muthyala, P., Lehrheuer, B., Hofmeister, M., Vorholt, A., Schmitz, K., Pischinger, S., Leitner, W., Bardow, A. (2024). Towards carbon-neutral and clean propulsion in heavy-duty transportation with hydroformylated Fischer-Tropsch fuels. Nature Energy, 9 (10), 1220-1229. doi:10.1038/s41560-024-01581-z.
- Thanheuser, N., Groteguth, J. T., Leitner, W., Esteban, J., Vorholt, A. J. (2024). Biphasic Production of 5-hydroxymethylfurfural (HMF) in a Recyclable Deep Eutectic Solvent-based System Catalyzed by H4SiW12O40. Chemsuschem. doi:10.1002/cssc.202401485.
- Stahl, S., Wessel, N., Vorholt, A. J., Leitner, W. (2024). Liquid-phase hydrogenation of carbon monoxide to methanol using a recyclable manganese-based catalytic system. Green Chemistry, 26 (13), 7799-7805. doi:10.1039/d4gc01050g
- Solmi, M. V., Vossen, J. T., Schmitz, M., Vorholt, A. J., Leitner, W. (2024). Catalytic synthesis of carboxylic acids from oxygenated substrates using CO2 and H2 as C1 building blocks. Green Chemistry, 26 (12), 7302-7311. doi:10.1039/d4gc01732c.
- Schoofs, L., Weidener, D., Leitner, W., Klose, H., Grande, P. M. (2024). Lignocellulose Treatment Using a Flow-Through Variant of OrganoCat Process. Chemsuschem. doi:10.1002/cssc.202401063.
- Nasse, K. E., Heinen, F. S., Pawlowsky, N., Schrimpf, M., Monflier, E., Tilloy, S., Leitner, W., Vorholt, A. J. (2024). The role of cyclodextrins in the acceleration of the reaction rate in a biphasic hydroformylations. Chemical Engineering Journal, 497, 154114. doi:10.1016/j.cej.2024.154114.
- Mondal, T., Leitner, W., Hölscher, M. (2024). Computational design of cooperatively acting molecular catalyst systems: carbene based tungsten- or molybdenum-catalysts with rhodium- or iridium-complexes for the ionic hydrogenation of N2 to NH3. Dalton Transactions, 53 (18), 7890-7898. doi:10.1039/d4dt00563e.
- Leitner, W. (2024). Carbon dioxide and hydrogen as building blocks for a sustainable interface of energy and chemistry. Philosophical Transactions of the Royal Society a-Mathematical Physical and Engineering Sciences, 382 (2282). doi:10.1098/rsta.2023.0266.
- Köhnke, K., Björnsson, R., Leitner, W., Vorholt, A. J., Mechanistic Aspects of Rhodium-Catalyzed Isoprene Hydroformylation: A Computational Study, Organometallics 2024. doi: 10.1021/acs.organomet.3c00414.
- Klos, N., Osterthun, O., Mengers, H. G., Lanzerath, P., von Westarp, W. G., Lim, G., Gausmann, M., Küsters-Spöring, J. D., Wiesenthal, J., Guntermann, N., Lauterbach, L., Jupke, A., Leitner, W., Blank, L. M., Klankermayer, J., Rother, D. (2024). Concatenating Microbial, Enzymatic, and Organometallic Catalysis for Integrated Conversion of Renewable Carbon Sources. Jacs Au. doi:10.1021/jacsau.4c00511.
- Kick, A. C., Weyhermüller, T., Hölscher, M., Kaeffer, N., Leitner, W. (2024). Understanding Ligand Effects on Bielectronic Transitions: Chemo- and Electroreduction of Rhodium Bis(Diphosphine) Complexes to Low Oxidation States. Angewandte Chemie-International Edition, 63 (37). doi:10.1002/anie.202408356.
- Fassbach, T. A., Ji, J. M., Vorholt, A. J., Leitner, W. (2024). Recycling of Homogeneous Catalysts-Basic Principles, Industrial Practice, and Guidelines for Experiments and Evaluation. ACS Catalysis, 14 (9), 7289-7298. doi:10.1021/acscatal.4c01006.
- Ehmann, K. R., Ji, J. M., Dinsing, K., Maier, C. R., Vorholt, A. J., Leitner, W. (2024). Introducing a Second Liquid Phase in the Carbon Dioxide Hydrogenation to Formic Acid: Impact on Catalytic Conversion in Ru-Catalyzed Systems. ACS Sustainable Chemistry & Engineering, 12 (42), 15569-15577. doi:10.1021/acssuschemeng.4c05493.
- Belleflamme, M., Hommes, J., Dervisoglu, R., Bartalucci, E., Wiegand, T., Beine, A. K., Leitner, W., Vorholt, A. J. (2024). Catalytic Upgrading of Acetaldehyde to Acetoin Using a Supported N-Heterocyclic Carbene Catalyst. Chemsuschem, 17 (22). doi:10.1002/cssc.202400647.
2018-2023
- Zhang, Y. Y., El Sayed, S., Kang, L. Q., Sanger, M., Wiegand, T., Jessop, P. G., Debeer, S., Bordet, A., Leitner, W., Adaptive Catalysts for the Selective Hydrogenation of Bicyclic Heteroaromatics using Ruthenium Nanoparticles on a CO2-Responsive Support, Angewandte Chemie-International Edition 62, (48) 2023. doi: 10.1002/anie.202311427.
- Vossen, J. T., Leitner, W., Vorholt, A. J., Exploring the Hurdles in Thermomorphic Multicomponent Systems in the Rhodium-Catalyzed Multiphase Hydroformylation, ACS Sustainable Chemistry & Engineering 11, (28) 2023, 10462-10470. doi: 10.1021/ACSsuschemeng.3c01885.
- Vossen, J. T., Hülsken, N., Vorholt, A. J., Leitner, W., Recycling of a thermoresponsive "catalyst pill": separation of a molecular catalyst in solid ethylenecarbonate in various reactions, Green Chemistry 25, (7) 2023, 2872-2880. https://doi.org/10.1039/d2gc04822a.
- Sodreau, A., Zahedi, H. G., Dervisoglu, R., Kang, L. Q., Menten, J., Zenner, J., Terefenko, N., Debeer, S., Wiegand, T., Bordet, A., Leitner, W., A Simple and Versatile Approach for the Low-Temperature Synthesis of Transition Metal Phosphide Nanoparticles from Metal Chloride Complexes and P(SiMe3)3, Advanced Materials 35, (49) 2023. doi: 10.1002/adma.202306621.
- Singh, A., Kemper, G., Weyhermüller, T., Kaeffer, N., Leitner, W., Activated Mn-MACHO Complexes Form Stable CO2 Adducts, Chemistry-a European Journal 2023. https://doi.org/10.1002/chem.202303438.
- Nattermann, M., Wenk, S., Pfister, P., He, H., Lee, S. H., Szymanski, W., Guntermann, N., Zhu, F. Y., Nickel, L., Wallner, C., Zarzycki, J., Paczia, N., Gaissert, N., Franciò, G., Leitner, W., Gonzalez, R., Erb, T. J., Engineering a new-to-nature cascade for phosphate-dependent formate to formaldehyde conversion in vitro and in vivo, Nature Communications 14, (1) 2023. https://doi.org/10.1038/s41467-023-38072-w.
- Mengers, H. G., Guntermann, N., von Westarp, W. G., Jupke, A., Klankermayer, J., Blank, L. M., Leitner, W., Rother, D., Three Sides of the Same Coin: Combining Microbial, Enzymatic, and Organometallic Catalysis for Integrated Conversion of Renewable Carbon Sources, Chemie Ingenieur Technik 95, (4) 2023, 485-490. https://doi.org/10.1002/cite.202200169.
- Marchenko, N., Lacroix, L. M., Ratel-Ramond, N., Leitner, W., Bordet, A., Tricard, S., Bimetallic Fe FexPt100–x Nanoparticles Immobilized on Supported Ionic Liquid Phases as Hydrogenation and Hydrodeoxygenation Catalysts: Influence of the Metal Content on Activity and Selectivity, ACS Applied Nano Materials 6, (21) 2023, 20231-20239. https://doi.org/10.1021/acsanm.3c03996.
- Sodreau, A., Zahedi, H. G., Dervisoglu, R., Kang, L., Menten, J., Zenner, J., Terefenko, N., DeBeer, S., Wiegand, T., Bordet, A., Leitner, W. (2023). A Simple and Versatile Approach for the Low-Temperature Synthesis of Transition Metal Phosphide Nanoparticles from Metal Chloride Complexes and P(SiMe3)3. ADVANCED MATERIALS. 2306621 (1-9) doi:10.1002/adma.202306621.
- Levin, N., Goclik, L., Walschus, H., Antil, N., Bordet, A., Leitner, W. (2023). Decarboxylation and Tandem Reduction/Decarboxylation Pathways to Substituted Phenols from Aromatic Carboxylic Acids Using Bimetallic Nanoparticles on Supported Ionic Liquid Phases as Multifunctional Catalysts. JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, 145(41), 22845-22854. doi:10.1021/jacs.3c09290.
- Durin, G., Kaeffer, N., Leitner, W. (2023). Electrocatalytic hydrogenation of unsaturated organic compounds with molecular complexes: Mechanistic views. Current Opinion in Electrochemistry, (41): 101371, pp. 1-10. doi:10.1016/j.coelec.2023.101371.
- Zhang, Y., El Sayed, S., Kang, L., Sanger, M., Wiegand, T., Jessop, P. G., DeBeer, S., Bordet, A., Leitner, W. (2023). Adaptive Catalysts for the Selective Hydrogenation of Bicyclic Heteroaromatics using Ruthenium Nanoparticles on a CO2-Responsive Support. Angewandte Chemie, International Edition in English, (XX): e202311427, pp. x-xx. doi:10.1002/anie.202311427.
- Durin, G., Lee, M., Pogany, M. A., Weyhermüller, T., Kaeffer, N., Leitner, W. (2023). Hydride-Free Hydrogenation: Unraveling the Mechanism of Electrocatalytic Alkyne Semihydrogenation by Nickel-Bipyridine Complexes. JOURNAL OF THE AMERICAN CHEMICAL SOCIETy, 145(31), 17103-17111. doi:10.1021/jacs.3c03340.
- Jenthra, S., Mondal, T., Kemper, G., Lantzius-Beninga, M., Hölscher, M., Leitner, W. (2023). Ligand-Controlled Palladium-Catalyzed Decarboxylative Heck Coupling for Regioselective Access to Branched Olefins. ACS Catalysis, (13), 10085-10093. doi:10.1021/acscatal.3c02224.
- Vossen, J. T., Leitner, W., Vorholt, A. J. (2023). Exploring the Hurdles in Thermomorphic Multicomponent Systems in the Rhodium-Catalyzed Multiphase Hydroformylation. ACS Sustainable Chemistry & Engineering, (11), 10462-10470. doi:10.1021/acssuschemeng.3c01885.
- Bordet, A., Leitner, W. (2023). Adaptive Catalytic Systems for Chemical Energy Conversion. Angewandte Chemie, International Edition in English, (62): E202301956, pp. 1-8. doi:10.1002/anie.202301956.
- Vossen, J. T., Hülsken, N., Vorholt, A. J., Leitner, W. (2023). Recycling of a thermoresponsive "catalyst pill": separation of a molecular catalyst in solid ethylene carbonate in various reactions. Green Chemistry, (25), 2872-2880. doi:10.1039/d2gc04822a.
- Kemper, G., Hölscher, M., Leitner, W. (2023). Pd(II)-catalyzed carboxylation of aromatic C?H bonds with CO2. Science Advances, 9(5): eadf2966, pp. 1-10. doi:10.1126/sciadv.adf2966.
- Cramer, H. H., Das, S., Wodrich, M. D., Corminboeuf, C., Werlé, C., Leitner, W. (2023). Theory-guided development of homogeneous catalysts for the reduction of CO2 to formate, formaldehyde, and methanol derivatives. Chemical Science, (14) 2799-2807. doi:10.1039/d2sc06793e. doi:10.1039/d2sc06793e.
- Anandaraj, S. J. L., Kang, L., DeBeer, S., Bordet, A., Leitner, W. (2023). Catalytic Hydrogenation of CO2 to Formate Using Ruthenium Nanoparticles Immobilized on Supported Ionic Liquid Phases. Small, (19)2206806, pp. 1-10. doi:10.1002/smll.202206806.
- Han, C., Zenner, J., Johny, J., Kaeffer, N., Bordet, A., Leitner, W. (2023). Electrocatalytic hydrogenation of alkenes with Pd/carbon nanotubes at an oil-water interface. Nature Catalysis, 5(12), 1110-1119. doi:10.1038/s41929-022-0088
- Ullmann, L., Guntermann, N., Kohl, P., Schröders, G., Müsgens, A., Francio, G., Leitner, W., Blank, L. M. (2022). Improved Itaconate Production with Ustilago cynodontis via Co-Metabolism of CO2-Derived Formate. JOURNAL OF FUNGI, 8(12): 1277, pp. 1-17. doi:10.3390/jof8121277.
- Mengers, H. G., Guntermann, N., von Westarp, W. G., Jupke, A., Klankermayer, J., Blank, L. M., Leitner, W., Rother, D. (2023). Three Sides of the Same Coin: Combining Microbial, Enzymatic, and Organometallic Catalysis for Integrated Conversion of Renewable Carbon Sources. Chemie-Ingenieur-Technik, 95(4),485-490 doi:10.1002/cite.202200169.
- Antico, E., Leutzsch, M., Wessel, N., Weyhermüller, T., Werlé, C., Leitner, W. (2022). Selective oxidation of silanes into silanols with water using [MnBr(CO)(5)] as a precatalyst. Chemical Science, 14(1), 54-60. doi:10.1039/d2sc05959b.
- Kalsi, D., Anandaraj, S. J. L., Durai, M., Weidenthaler, C., Emondts, M., Nolan, S. P., Bordet, A., Leitner, W. (2022). One-Pot Multicomponent Synthesis of Allyl and Alkylamines Using a Catalytic System Composed of Ruthenium Nanoparticles on Copper N-Heterocyclic Carbene-Modified Silica. ACS Catalysis, 12(24), 14902-14910. doi:10.1021/acscatal.2c04044.
- Kuss, D. A., Hölscher, M., Leitner, W. (2022). Combined Computational and Experimental Investigation on the Mechanism of CO2 Hydrogenation to Methanol with Mn-PNP-Pincer Catalysts. ACS Catalysis, (12), 15310-15322. doi:10.1021/acscatal.2c04806.
- Diehl, T., Lanzerath, P., Francio, G., Leitner, W. (2022). A Self-Separating Multiphasic System for Catalytic Hydrogenation of CO2 and CO2-Derivatives to Methanol. ChemSusChem, (15): e202201250, pp. 1-8. doi:10.1002/cssc.202201250.
- October, J., Köhnke, K., Thanheuser, N., Vorholt, A. J., Leitner, W. (2022). Reppe-Carbonylation of Alkenes with Carboxylic Acids: A Catalytic and Mechanistic Study. European Journal of Organic Chemistry, 2022(43): e202201018, pp. 1-7. doi:10.1002/ejoc.202201018.
- Ehmann, K. R., Nisters, A., Vorholt, A. J., Leitner, W. (2022). Carbon Dioxide Hydrogenation to Formic Acid with Self-Separating Product and Recyclable Catalyst Phase. ChemCatChem, 14(19): e2022008, pp. 1-7. doi:10.1002/cctc.202200892.
- Guntermann, N., Francio, G., Leitner, W. (2022). Hydrogenation of CO2 to formic acid in biphasic systems using aqueous solutions of amino acids as the product phase. Green Chemistry, (xx), 1-7. doi:10.1039/d2gc02598a.
- Rösler, T., Betting, J., Püschel, S., Vorholt, A. J., Leitner, W. (2022). Solvent design for catalyst recycling of rhodium/amine catalysts via scCO(2) extraction in the reductive hydroformylation of alpha olefins. Green Chemistry, 24(17), 6578-6588. doi:10.1039/d2gc01252a.
- Strohmann, M. P., Vorholt, A. J., Leitner, W. (2022). Branched Tertiary Amines from Aldehydes and alpha-Olefins by Combined Multiphase Tandem Reactions. CHEMISTRY A EUROPEAN JOURNAL, (xx): e202202081, pp. 1-8. doi:10.1002/chem.202202081.
- Lin, S.-H., Hetaba, W., Chaudret, B., Leitner, W., Bordet, A. (2022). Copper-Decorated Iron Carbide Nanoparticles Heated by Magnetic Induction as Adaptive Multifunctional Catalysts for the Selective Hydrodeoxygenation of Aldehydes. Advanced Energy Materials,12(42) 2201783, pp. 1-10. doi:10.1002/aenm.202201783.
- Kliemann, M. N., Teeuwen, S., Weike, C., Francio, G., Leitner, W. (2022). Rhodium-Catalyzed Asymmetric Hydrohydrazonemethylation of Styrenes: Access to Chiral Hydrazones, Hydrazides, Hydrazines and Amines. Advanced Synthesis & Catalysis, (364), 4006-4012. doi:10.1002/adsc.202200804.
- Kacem, S., Qiao, Y., Wirtz, C., Theyssen, N., Bordet, A., Leitner, W. (2022). Supercritical carbon dioxide as reaction medium for selective hydrogenation of fluorinated arenes. Green Chemistry, (24), 8671-8676. doi:10.1039/d2gc02623f..
- Jürling-Will, P., Botz, T., Francio, G., Leitner, W. (2022). A "Power-to-X" Route to Acetic Acid via Palladium-Catalyzed Isomerization of Methyl Formate. ChemSusChem, e202201006, pp. 1-7. doi:10.1002/cssc.202201006.
- Schrimpf, M., Graefe, P. A., Holl, A., Vorholt, A. J., Leitner, W. (2022). Effect of Liquid-Liquid Interfacial Area on Biphasic Catalysis Exemplified by Hydroformylation. ACS Catalysis, (12), 7850-7861. doi:10.1021/acscatal.2c01972.
- Kaeffer, N. Leitner, W. (2022). Electrocatalysis with Molecular Transition-Metal Complexes for Reductive Organic Synthesis. JACS Au 2022 2 (6), 1266-1289. doi:10.1021/jacsau.2c00031
- Jeske, K., Rösler, T., Belleflamme, M., Rodenas, T., Fischer, N., Claeys, M., Leitner, W., Vorholt, A. J., Prieto, G. (2022). Direct Conversion of Syngas to Higher Alcohols via Tandem Integration of Fischer-Tropsch Synthesis and Reductive Hydroformylation. Angewandte Chemie, International Edition in English, e202201004, pp. 1-9. doi:10.1002/anie.202201004.
- Vossen, J. T., Vorholt, A. J., Leitner, W. (2022). Catalyst Recycling in the Reactive Distillation of Primary Alcohols to Olefins Using a Phosphoric Acid Catalyst. ACS SUSTAINABLE CHEMISTRY & ENGINEERING, 10(18), 5922-5931. doi:10.1021/acssuschemeng.2c00237.
- Lee, M.-Y., Kahl, C., Kaeffer, N., Leitner, W. (2022). Electrocatalytic Semihydrogenation of Alkynes with [Ni(bpy)3]2+. Journal of the American Chemical Society, (2), 573-578. doi:10.1021/jacsau.1c00574.
- Sisodiya-Amrute, S., Van Stappen, C., Rengshausen, S., Han, C., Sodreau, A., Weidenthaler, C., Tricard, S., DeBeer, S., Chaudret, B., Bordet, A., Leitner, W. (2022). <p>Bimetallic MxRu100_x nanoparticles (M = Fe, Co) on supported ionic liquid phases (MxRu100-x@SILP) as hydrogenation catalysts: Influence of M and M:Ru ratio on activity and selectivity</p>. Journal of Catalysis,(407), 141-148. doi:10.1016/j.jcat.2022.01.030.
- Leitner, W. (2022). If sustainability is the goal, green chemistry will show the way! - happy birthday to Paul Anastas. Green Chemistry, 24(9), 3374-3375. doi:10.1039/d2gc90035a.
- Köhnke, K., Wessel, N., Esteban, J., Jing, J., Vorholt, A. J., Leitner, W. (2022). Operando monitoring of mechanisms and deactivation of molecular catalysts. Green Chemistry, 24(5), 1951-1972. doi:10.1039/d1gc04383h.
- Hoffmann, M., Hermesmann, M., Leven, M., Leitner, W., Müller, T. E. (2022). Semi-Crystalline Polyoxymethylene-co-Polyoxyalkylene Multi-Block Telechels as Building Blocks for Polyurethane Applications. Polymers, 14(5): 882, pp. 1-24. doi:10.3390/polym14050882.
- Goclik, L., Walschus, H., Bordet, A., Leitner, W. (2022). Selective hydrodeoxygenation of acetophenone derivatives using a Fe25Ru75@SILP catalyst: a practical approach to the synthesis of alkyl phenols and anilines. Green Chemistry, (24), 2937-2945. doi:10.1039/d1gc04189d.
- Hölscher, M., Kemper, G., Jenthra, S., Bolm, C., Leitner, W. (2022). Factors Governing the Catalytic Insertion of CO2 into Arenes - A DFT Case Study for Pd and Pt Phosphane Sulfonamido Complexes. Chemistry – A European Journal, e202104375, pp. 1-9. doi:10.1002/chem.202104375.
- Machat, M. R., Marbach, J., Schumacher, H., Raju, S., Lansing, M., Over, L. C., Adler, L., Langanke, J., Wolf, A., Leitner, W., Gürtler, C. (2022). Turning CO/CO2-containing industrialprocess gas into valuable building blocks for the polyurethane industry. REACTION CHEMISTRYENGINEERING, (7), 580-589. doi:10.1039/d1re00508a.
- Püschel, S., Hammami, E., Rösler, T., Ehmann, K. R., Vorholt, A. J., Leitner, W. (2022). Auto-tandem catalytic reductive hydroformylation with continuous multiphase catalyst recycling. Catalysis Science & Technology, (12) 728-736. doi:10.1039/d1cy02000e.
- Guntermann, N., Mengers, H. G., Francio, G., Blank, L. M., Leitner, W. (2021). Bio-energy conversion with carbon capture and utilization (BECCU): integrated biomass fermentation and chemo-catalytic CO2 hydrogenation for bioethanol and formic acid co-production. Green Chemistry, 23(24), 9860-9864. doi:10.1039/d1gc02915k.
- Cramer, H. H., Ye, S., Neese, F., Werlé, C., Leitner, W. (2021). Cobalt-Catalyzed Hydrosilylation of Carbon Dioxide to the Formic Acid, Formaldehyde, and Methanol Level-How to Control the Catalytic Network? JACS Au, 1(11), 2058-2069. doi:10.1021/jacsau.1c00350.
- Kinzel, N. W., Demirbas, D., Bill, E., Weyhermüller, T., Werlé, C., Kaeffer, N., Leitner, W. (2021). Systematic Variation of 3d Metal Centers in a Redox-Innocent Ligand Environment: Structures, Electrochemical Properties, and Carbon Dioxide Activation. Inorganic Chemistry, (xx), xx-xx. doi:10.1021/acs.inorgchem.1c02909.
- Kinzel, N. W., Werlé, C., Leitner, W. (2021). Transition Metal Complexes as Catalysts for the Electroconversion of CO2: An Organometallic Perspective. Angewandte Chemie, International Edition in English, (60), 11268-11686. https://doi.org/10.1002/anie.202006988
- Kreissl, H., Jin, J., Lin, S.-H., Schütte, D., Störtte, S., Levin, N., Chaudret, B., Vorholt, A., Bordet, A. Leitner, W. (2021). Commercial Cu2Cr2O5 Decorated with Iron Carbide Nanoparticles as Multifunctional Catalyst for Magnetically Induced Continuous Flow Hydrogenation of Aromatic Ketones. Angewandte Chemie, International Edition in English, (xx), xx-xx. doi:10.1002/anie.202107916.
- Kaithal, A., Chatterjee, B., Werlé, C., Leitner, W. (2021). Acceptorless dehydrogenation of methanol to carbon monoxide and hydrogen using molecular catalysts. Angewandte Chemie, International Edition in English, (xx), xx-xx. doi:10.1002/anie.202110910.
- Zenner, J., Moos, G., Luska, K. L., Bordet, A., Leitner, W. (2021). Rh NPs Immobilized on Phosphonium- based Supported Ionic Liquid Phases (Rh@SILPs) as Hydrogenation Catalysts. Chimia, 75(9), 724-732. doi:10.2533/chimia.2021.724.
- Weidener, D., Klose, H., Graf von Westarp, W., Jupke, A., Leitner, W., De Maria, P. D., Grande,P.M. (2021). Selective lignin fractionation using CO2-expanded 2-methyltetrahydrofuran (2-MTHF). Green Chemistry, (23), 6330-6336. doi:10.1039/D1GC01651B.
- Rösler, T., Ehmann, K. R., Köhnke, K., Leutzsch, M., Wessel, N., Vorholt, A. J., Leitner, W. (2021). Reductive hydroformylation with a selective and highly active rhodium amine system. Journal of Catalysis, 400, 234-243. doi:10.1016/j.jcat.2021.06.001.
- Bardow, A., Bizzarri, C., Cao, X. E., Cowan, A. J., Cummings, C., Del Angel Hernandez, V., Doan, H., Dowson, G., Ghosh, S., Gil, V., Gugujonovic, K., Kamali, A. R., König, M., Leitner, W ., Luo, J., et.al.(2021). Emerging technologies: general discussion. Faraday Discussions, 230(0), 388-412. doi:10.1039/d1fd90048j.
- Antico, E., Schlichter, P., Werle, C., Leitner, W. (2021) Reduction of Carboxylic Acids to Alcohols via Manganese(I) Catalyzed Hydrosilylation. JACS AU, 1(6), 742-749. doi:10.1021/jacsau.1c00140.
- Bordet, A., El Sayed, S., Sanger, M., Boniface, K. J., Kalsi, D., Luska, K. L., Jessop, P.; Leitner,W. (2021). Selectivity control in hydrogenation through adaptive catalysis using ruthenium nanoparticles on a CO2-responsive support. Nature chemistry. doi:10.1038/s41557-021-00735-w.
- Leitner, W., & Schmitz, M. (2021). Concluding remarks: Carbon dioxide utilization: where are we now? and where are we going? Faraday discussions.(XX) XX-XX doi:10.1039/d1fd00038a.
- Püschel, S., Störtte, S., Topphoff, J., Vorholt, A. J., Leitner, W. (2021). Green process design for reductive hydroformylation of renewable olefin cuts for drop-in diesel fuels. ChemSusChem, (14), 5226-5234 . doi:10.1002/cssc.202100929.
- Bordet, A., Leitner, W. (2021). Metal Nanoparticles Immobilized on Molecularly Modified Surfaces: Versatile Catalytic Systems for Controlled Hydrogenation and Hydrogenolysis. Accounts of Chemical Research, (xxx), xxx-xxx. doi:10.1021/acs.accounts.1c00013.
- Kaithal, A., Hölscher, M., Leitner, W. (2021). Carbon monoxide and hydrogen (syngas) as a C1-building block for selective catalytic methylation Chemical Science. 12, 976-982 https://doi.org/10.1039/D0SC05404F
- Kuß, D. A., Hölscher, M., Leitner, W. (2021). Hydrogenation of CO2 to Methanol with Mn-PNP-Pincer Complexes in the Presence of Lewis Acids: the Formate Resting State Unleashed. CHEMCATCHEM . (13) 1-6 doi:10.1002/cctc.202100649.
- Rengshausen, S., Van Stappen, C., Levin, N., Tricard, S., Luska, K.L., DeBeer, S., Chaudret, B., Bordet, A., Leitner, W. (2021). Organometallic Synthesis of Bimetallic Cobalt‐Rhodium Nanoparticles in Supported Ionic Liquid Phases (CoxRh100−x@SILP) as Catalysts for the Selective Hydrogenation of Multifunctional Aromatic Substrates Small, 17,2006683 (10pp) https://doi.org/10.1002/smll.202006683
- Weidener, D, Leitner, W., Domínguez de María, P., Klose, H., Grande, P.M. (2021). Lignocellulose fractionation using recyclable phosphoric acid: Lignin, cellulose and furfural production. ChemSusChem. 14, 909-916, https://doi.org/10.1002/cssc.202002383
- Kaithal, A., Kalsi, D., Krishnakumar, V., Pattanaik, S., Bordet, A., Leitner, W., Gunanathan, C. (2020). Ruthenium-Catalyzed Selective Hydroboronolysis of Ethers ACS Catalysis 10(24), 14390-14397. https://doi.org/10.1021/acscatal.0c04269
- Voit, G., Jenthra, S., Hölscher, M., Weyhermüller, T., Leitner, W. (2020). Reversible Insertion of Carbon Dioxide at Phosphine Sulfonamido PdII–Aryl Complexes Organometallics 39(24), 4465-4473. https://doi.org/10.1021/acs.organomet.0c00560
- Bordet, A., Moos, G., Welsh, C., Licence, P., Luska K.L., Leitner, W. (2020). Molecular Control of the Catalytic Properties of Rhodium Nanoparticles in Supported Ionic Liquid Phase (SILP) Systems ACS Catalysis 10(23), 13904-13912. https://doi.org/10.1021/acscatal.0c03559
- Strohmann, M., Vossen, J.T., Vorholt, A.J., Leitner, W. (2020). Recycling of two molecular catalysts in the hydroformylation/aldol condensation tandem reaction using one multiphase system Green Chemistry 22(23), 8444-8451. https://doi.org/10.1039/D0GC03392H
- Kacem, S., Emondts, M., Bordet, A., Leitner, W. (2020). Selective hydrogenation of fluorinated arenes using rhodium nanoparticles on molecularly modified silica Catalysis Science & Technology 10(23), 8120-8126. https://doi.org/10.1039/D0CY01716G
- Pandey, P., Daw, P., Din Reshi, N.U., Ehmann, K.R., Hölscher, M., Leitner, W., Bera, J.K. (2020). A Proton-Responsive Annulated Mesoionic Carbene (MIC) Scaffold on Ir Complex for Proton/Hydride Shuttle: An Experimental and Computational Investigation on Reductive Amination of Aldehyde Organometallics 39(21), 3849-3863. https://doi.org/10.1021/acs.organomet.0c00568
- Weidener, D., Dama, M., Dietrich, S.K., Ohrem, B., Pauly, M., Leitner, W., Domínguez de Maria, P., Grande, P.M., Klose, H. (2020). Multiscale analysis of lignocellulose recalcitrance towards OrganoCat pretreatment and fractionation Biotechnology for Biofuels 13, 155. https://doi.org/10.1186/s13068-020-01796-8
- Hussong, C., Langanke, J., Leitner, W. (2020). A green route to polyurethanes: oxidative carbonylation of industrially relevant aromatic diamines by CO2-based methyl formate Green Chemistry 22(23), 8260-8270. https://doi.org/10.1039/D0GC02412K
- Hussong, C., Langanke, J., Leitner, W. (2020). Carbon2Polymer: A CO2‐based Route to Polyurethanes via Oxidative Carbonylation of TDA with Methyl Formate Chemie Ingenieur Technik 92(10), 1482-1488. https://doi.org/10.1002/cite.202000031
- Goclik, L., Offner-Marko, L., Bordet, A., Leitner, W. (2020). Selective Hydrodeoxygenation of Hydroxyacetophenones to Ethyl-Substituted Phenol Derivatives Using a FeRu@SILP Catalyst Chemical Communications 56(66), 9509-9512. https://doi.org/10.1039/D0CC03695A
- Weidener, D., Holtz, A., Klose, H., Jupke, A., Leitner, W., Grande, P.M. (2020). Lignin Precipitation and Fractionation from OrganoCat Pulping to Obtain Lignin with Different Sizes and Chemical Composition Molecules 25(15), 3330. https://doi.org/10.3390/molecules25153330
- Chatterjee, B., Kalsi, D., Kaithal, A., Bordet, A., Leitner, W., Gunanathan, C. (2020). One-pot dual catalysis for the hydrogenation of heteroarenes and arenes Catalysis Science & Technology. https://doi.org/10.1039/D0CY00928H
- Beck-Sickinger, A.G., Carell, T., Dehnen, S., Leitner, W., Schreiner, P.R., Wennemers, H. (2020). From Scientists to Scientists—Moving Angewandte into the Future Angewandte Chemie International Edition 59(31), 12548-12549. https://doi.org/10.1002/anie.202008469
- Cramer, H.H., Chatterjee, B., Weyhermüller, T., Werlé, C., Leitner, W. (2020). Controlling the Product Platform of Carbon Dioxide Reduction: Adaptive Catalytic Hydrosilylation of CO2 Using a Molecular Cobalt(II) Triazine Complex Angewandte Chemie International Edition 59(36), 15674-15681. https://doi.org/10.1002/anie.202004463
- Erken, C., Hindemith, C., Weyhermüller, T., Hölscher, M., Werlé, C., Leitner, W. (2020). Hydroamination of Aromatic Alkynes to Imines Catalyzed by Pd(II)–Anthraphos Complexes ACS Omega 5(15), 8912-8918. https://doi.org/10.1021/acsomega.0c00562
- Moos, G., Emondts, M., Bordet, A., Leitner, W. (2020). Selective Hydrogenation and Hydrodeoxygenation of Aromatic Ketones to Cyclohexane Derivatives Using a Rh@SILP Catalyst Angewandte Chemie International Edition 59(29), 11977-11983. https://doi.org/10.1002/anie.201916385
- Esteban, J., Vorholt, A.J., Leitner, W. (2020). An overview of the biphasic dehydration of sugars to 5-hydroxymethylfurfural and furfural: a rational selection of solvents using COSMO-RS and selection guides Green Chemistry. https://doi.org/10.1039/C9GC04208C
- Estes, D.P., Leutzsch, M., Schubert, L., Bordet, A., Leitner, W. (2020). The Effect of Ligand Electronics on the Reversible Catalytic Hydrogenation of CO2 to Formic Acid using Ruthenium Polyhydride Complexes: A Thermodynamic and Kinetic Study ACS Catalysis 10(5), 2990-2998. https://doi.org/10.1021/acscatal.0c00404
- Zimmerman, J.B., Anastas, P.T., Erythropel, H.C., Leitner, W. (2020). Designing for a green chemistry future Science 367(6476), 397-400. https://doi.org/10.1126/science.aay3060
- El Sayed, S., Bordet, A., Weidenthaler, C., Hetaba, W., Luska, K., Leitner, W. (2020) Selective Hydrogenation of Benzofurans using Lewis Acid Modified Ruthenium-SILP Catalysts ACS Catalysis 10(3), 2124-2130. https://doi.org/10.1021/acscatal.9b05124
- Holtz, A., Weidener, D., Leitner, W., Klose, H., Grande, P.M., Jupke, A. (2020). Process development for separation of lignin from OrganoCat lignocellulose fractionation using antisolvent precipitation Separation and Purification Technology 236, 116295. https://doi.org/10.1016/j.seppur.2019.116295
- Kaithal, A., van Bonn, P., Hölscher, M., Leitner, W. (2020). Manganese(I)‐Catalyzed β‐Methylation of Alcohols using Methanol as C1 Source Angewandte Chemie International Edition 59(1), 215-220. https://doi.org/10.1002/anie.201909035
- Kaithal, A., Schmitz, M., Hölscher, M., Leitner, W. (2020). On the mechanism of the Ruthenium‐catalyzed β‐methylation of alcohols with methanol ChemCatChem 12(3), 781-787. https://doi.org/10.1002/cctc.201901871
- Scott, M., Westhues, C.G., Kaiser, T., Baums, J.C., Jupke, A., Franciò, G., Leitner, W. (2019). Methylformate from CO2: an integrated process combining catalytic hydrogenation and reactive distillation Green Chemistry 21(23), 6307-6317. https://doi.org/10.1039/c9gc03006a
- Strohmann, M., Bordet, A., Vorholt, A.J., Leitner, W. (2019). Tailor-Made Biofuel 2-Butyltetrahydrofuran from the Continuous Flow Hydrogenation and Deoxygenation of Furfuralacetone Green Chemistry 21(23), 6299-6306. https://doi.org/10.1039/c9gc02555c
- Kaithal, A., Gracia, L.-L., Camp, C., Quadrelli, E.A., Leitner, W. (2019). Direct Synthesis of Cycloalkanes from Diols and Secondary Alcohols or Ketones Using a Homogeneous Manganese Catalyst Journal of the American Chemical Society 141(44), 17487-17492. https://doi.org/10.1021/jacs.9b08832
- Martínez-Ferraté, O., Chatterjee, B., Werlé, C., Leitner, W. (2019). Hydrosilylation of carbonyl and carboxyl groups catalysed by Mn(I) complexes bearing triazole ligands Catalysis Science & Technology 9(22), 6370-6379. https://doi.org/10.1039/c9cy01738k
- Grande, P.M., Weidener, D., Dietrich, S., Dama, M., Bellof, M., Maas, R., Pauly, M., Leitner, W., Klose, H., Domínguez de María, P. (2019). OrganoCat Fractionation of Empty Fruit Bunches from Palm Trees into Lignin, Sugars, and Cellulose-Enriched Pulp ACS Omega 4(11), 14451-14457. https://doi.org/10.1021/acsomega.9b01371
- Rohner, S.S., Kinzel, N., Werlé, C., Leitner, W. (2019). Systematic ligand variation to modulate the electrochemical properties of iron and manganese complexes Dalton Transactions 48, 13205-13211. https://doi.org/10.1039/c9dt01343a
- Liu, H., Zhu, L., Wallraf, A.M., Räuber, C., Grande, P.M., Anders, N., Gertler, C., Werner, B., Klankermayer, J., Leitner, W., Schwaneberg, U. (2019). Depolymerization of laccase-oxidized lignin in aqueous alkaline solution at 37°C ACS Sustainable Chemistry & Engineering 7(13), 11150-11156. https://doi.org/10.1021/acssuschemeng.9b00204
- Kaithal, A., Schmitz, M., Hölscher, M., Leitner, W. (2019). Ruthenium(II)-Catalyzed ß-Methylation of Alcohols using Methanol as C1 Source 11(21), 5287-5291. ChemCatChem. https://doi.org/10.1002/cctc.201900788
- Schrimpf, M., Esteban, J., Rösler, T., Vorholt, A.J., Leitner, W. (2019). Intensified Reactors for Gas-Liquid-Liquid Multiphase Catalysis: from Chemistry to Engineering Chemical Engineering Journal 372, 917-939. https://doi.org/10.1016/j.cej.2019.03.133
- Rengshausen, S., Etscheidt, F., Großkurth, J., Luska, K.L., Bordet, A., Leitner, W. (2019). Catalytic Hydrogenolysis of Substituted Diaryl Ethers by Using Ruthenium Nanoparticles on an Acidic Supported Ionic Liquid Phase (Ru@SILP-SO3H) Synlett 30(04), 405-412. https://doi.org/10.1055/s-0037-1611678
- Jens, C.M., Scott, M., Liebergesell, B., Westhaus, C.G., Schäfer, P., Franciò, G., Leonhard, K., Leitner, W., Bardow, A. (2019). Rh-Catalyzed Hydrogenation of CO2 to Formic Acid in DMSO-based Reaction Media: Solved and Unsolved Challenges for Process Development Advanced Synthesis and Catalysis 361(2), 307-316. https://doi.org/10.1002/adsc.201801098
- Faßbach, T.A., Vorholt, A.J., Leitner, W. (2019). The Telomerization of 1,3 Dienes – A Reaction Grows Up ChemCatChem 11(4), 1153-1166. https://doi.org/10.1002/cctc.201801821
- Rösler, T., Faβbach, T.A., Schrimpf, M., Vorholt, A.J., Leitner, W. (2019). Towards water-based recycling techniques: Methodologies for homogeneous catalyst recycling in liquid/liquid multiphase media and their implementation in continuous processes Industrial & Engineering Chemistry Research 58(7), 2421-2436. https://doi.org/10.1021/acs.iecr.8b04295
- Solmi, M.V., Schmitz, M., Leitner, W. (2019). CO2 as a Building Block for the Catalytic Synthesis of Carboxylic Acids Studies in Surface Science and Catalysis: Horizons in Sustainable Industrial Chemistry and Catalysis 178, Chapter 6, 105-124. https://doi.org/10.1016/B978-0-444-64127-4.00006-9
- Cantat, T., Leitner, W. (2019). Section 6: Homogeneous catalysis for the sustainable production of fuels and chemicals Research needs: Towards sustainable production of fuels and chemicals, EnergyX Report 60-68. https://www.energy-x.eu/wp-content/uploads/2019/09/Energy_X_Research-needs-report.pdf
- Erken, C., Kaithal, A., Sen, S., Weyhermüller, T., Hölscher, M., Werlé, C., Leitner, W. (2018). Manganese-catalyzed hydroboration of carbon dioxide and other challenging carbonyl groups Nature Communications 9, 4521. https://doi.org/10.1038/s41467-018-06831-9
- Regenstein, L., Klement, T., Grande, P., Kryenschulte, D., Heyman, B., Maßmann, T., Eggert, A., Sengspiel, R., Wang, Y., Wierckx, N., Blanck, L.M., Spiess, A., Leitner, W., Bolm, C., Wessling, M., Jupke, A., Rosenbaum, M., Büchs, J. (2018). From beech wood to itaconic acid: case study on biorefinery process integration Biotechnology for Biofuels 11, 279-289. https://doi.org/10.1186/s13068-018-1273-y
- Schmitz, M., Erken, C., Ohligschläger, A., Schnoor, J.-K., Westhues, N.F., Klankermayer, J., Leitner, W., Liauw, M.A. (2018). Homogeneously Catalyzed Synthesis of (Higher) Alcohols (C1–C4) from the Combination of CO2/CO/H2 Chemie Ingenieur Technik 90(10), 1476-1488. https://doi.org/10.1002/cite.201800053
- Martínez-Ferraté, O., Werlé, C., Franciò, G., Leitner, W. (2018). Aminotriazole Mn(I) Complexes as Effective Catalysts for Transfer Hydrogenation of Ketones ChemCatChem 23, 4514-4518. https://doi.org/10.1002/cctc.201800953
- Offner-Marko, L., Bordet, A., Moos, G., Tricard, S., Rengshausen, S., Chaudret, B., Luska, K.L., Leitner, W. (2018). Bimetallic Nanoparticles in Supported Ionic Liquid Phases as Multifunctional Catalysts for the Selective Hydrodeoxygenation of Aromatic Substrates Angewandte Chemie International Edition 57(39), 12721-12726. https://doi.org/10.1002/anie.201806638
- Kaithal, A., Hölscher, M., Leitner, W. (2018). Catalytic Hydrogenation of Cyclic Carbonates using Manganese Complexes Angewandte Chemie International Edition 57(41), 13449-13453. https://doi.org/10.1002/anie.201808676
- Leitner, W., Franciò, G., Scott, M., Westhues, C., Langanke, J., Lansing, M., Hussong, C., Erdkamp, E. (2018). Carbon2Polymer – Chemical Utilization of CO2 in the Production of Isocyanates Chemie Ingenieur Technik 90(10), 1504-1512. https://doi.org/10.1002/cite.201800040
- Dutta, I., Yadav, S., Sarbajna, A., De, S., Hölscher, M., Leitner, W., Bera, J.K. (2018). Double Dehydrogenation of Primary Amines to Nitriles by a Ruthenium Complex Featuring Pyrazole Functionality Journal of the American Chemical Society 140(28), 8662-8666. https://doi.org/10.1021/jacs.8b05009
- Pienkoß, F., Ochoa-Hernández, Theyssen, N., Leitner, W. (2018). Kaolin: A Natural Low-Cost Material as Catalyst for Isomerization of Glucose to Fructose ACS Sustainable Chemistry & Engineering 6(7), 8782-8789. https://doi.org/10.1021/acssuschemeng.8b01151
- Weidener, D., Klose, H., Leitner, W., Schurr, U., Usadel, B., Domínguez de Maria, P., Grande, P.M. (2018). One-step lignocellulose fractionation using 2,5-furandicarboxylic acid as biogenic and recyclable catalyst ChemSusChem 11(13), 2051-2056. https://doi.org/10.1002/cssc.201800653
- Deutz, S., Bongartz, D., Heuser, B., Katelhon, A., Langenhorst, L. S., Omari, A., Walters, M., Diaeresis, S., Leitner, W., Mitsos, A., Pischinger, S., Bardow, A. (2018). Cleaner production of cleaner fuels: wind-to-wheel - environmental assessment of CO2-based oxymethylene ether as a drop-in fuel Energy Environmental Science 11, 331-343. https://doi.org/10.1039/C7EE01657C
- Artz, J., Müller, T.E., Thenert, K., Kleinekorte, J., Meys, R., Sternberg, A., Bardow, A., Leitner, W. (2018). Sustainable conversion of carbon dioxide: an integrated review of catalysis and life cycle assessment Chemical Reviews 118 (2), 434-504. https://doi.org/10.1021/acs.chemrev.7b00435
- Geier, D.I., Schmitz, P., Walkowiak, J., Leitner, W., Franciò, G. (2018). Continuous Flow Asymmetric Hydrogenation with Supported Ionic Liquid Phase Catalysts Using Modified CO2 as the Mobile Phase: from Model Substrate to an Active Pharmaceutical Ingredient ACS Catalysis 8, 3297-3303. https://doi.org/10.1021/acscatal.8b00216
- Schmitz, M., Solmi, M.V., Leitner, W. (2018). Catalytic Processes Combining CO2 and Alkenes into Value-Added Chemicals: Synthesis of Cyclic Carbonates, Lactones, Carboxylic Acids, Esters, Aldehydes, Alcohols, and Amines Organometallics for Green Catalysis, Topics in Organometallic Chemistry 63, 17-38. https://doi.org/10.1007/3418_2018_24
Lehrstuhl 'Technische Chemie und Petrolchemie' - RWTH Aachen
Prof. Leitner ist seit 2002 Lehrstuhlinhaber und Professor für Technische Chemie und Petrolchemie an der RWTH Aachen.
Webseite: RWTH Aachen - Lehrstuhl für Technische Chemie und Petrolchemie
Preise, Ehrungen & Auszeichnungen
- 2020 Prix Binational Franco-Allemand “Georg Wittig-Victor Grignard” awarded by the Société Chimique de France
- 2020 Top 3 of the European Innovator Award “Industry” together with Dr. Christoph Gürtler (Covestro AG)
- 2020 Top 10 Winners of the Falling Walls Breakthroughs of the Year in Physical Sciences
- 2019 Top 3 “Deutscher Zukunftspreis”, together with Dr. Christoph Gürtler and Dr. Berit Stange (Covestro AG)
- 2014 European Sustainable Chemistry Award of the European Science Association of Chemical and Molecular Sciences (EuCheMS), jointly with Prof. Jürgen Klankermayer (RWTH Aachen)
- 2009 Wöhler Award of the Gesellschaft Deutscher Chemiker (GDCh)
- 2008 CATSA Eminent Visitor Award of the South African Catalysis Society
- 2001 Otto-Roelen-Medal of DECHEMA
- 2000 2nd International Messer Innovation-Award
- 1998 Bennigsen-Foerder-Preis of Nordrhein-Westfalen (NRW)
- 1998 Carl-Zerbe-Award of Deutsche Wissenschaftliche Gesellschaft für Erdöl, Erdgas und Kohle (DMGK)
- 1997 Gerhard-Hess-Award of Deutsche Forschungsgemeinschaft (DFG)
Named Lectureships and Guest Professorships
- 2019 International Organic Chemistry Forum Lectureship, Kyoto/Osaka, Japan
- 2018 Casey Lecture of the University of Wisconsin, Madison, USA
- 2018 Evonik Lecture at the Biennial Meeting of the Chinese Chemical Society, Hangzhou, China
- 2017 Rennes Green Chemistry Lectureship, Rennes, France
- 2015 Nankai University Lectureship in Organic Chemistry, Nankai University, China
- 2015 Molecular Science Forum Lecture of Chinese Academy of Sciences and Chemical Society
- 2013 Visiting Lecturer for Promotion of Chemistry, National Science Foundation Taiwan
- 2005 Guest Professorship at the Universitè de Bourgogne, Dijon, France
- 2003 Griess Lectureship of the Royal Society of Chemistry (RSC)
Funktionen & Aufgaben
Journals
- Since 2021 Advisory Editor of Angewandte Chemie (GDCh, Wiley-VCH)
- 2018 - 2021 Member of the Kuratorium/Editorial Board of Angewandte Chemie (GDCh, Wiley-VCH)
- Since 2005 Member of the International Advisory Board of “Advanced Synthesis and Catalysis” (Wiley)
- 2004 - 2010 / 2010-2016 Scientific Editor / Chairman of the Editorial Board of “Green Chemistry” (Royal Society of Chemistry)
Conferences
- 2022 Chairman of Gordon Research Conference on Green Chemistry, Castelldefels, Spain
- 2019 Chairman of the 17th International Conference in Carbon Dioxide Utilization (ICCDU XVII), Aachen, Germany
- 2019 Chairman of the 14th European Congress on Catalysis (EuropaCat), Aachen, Germany
- Since 2018 Advisory Board of the International Symposium on Green Chemistry (ISGC), La Rochelle, France
- Since 2016 Advisory Board of the International Symposium on Homogeneous Catalysis (ISHC)
- 2012 Member of the Organizing Committee of the 15th International Congress on Catalysis (ICC), Munich, Germany
- 2002 - 2016 Chairman of the Conference Series “Green Solvents”
Coordinated Research Programs
- Since 2019 Co-Speaker of the Cluster of Excellence “The Fuel Science Center” at RWTH Aachen University and MPI CEC
- Since 2016 Chairman of the Coordination Group of the BMBF funded Kopernikus Project “P2X: Exploration, Validation, and Implementation of „Power-to-X“ Concepts”
- 2014 - 2018 Co-Speaker of the Cluster of Excellence “Tailor-Made Fuels from Biomass” at RWTH Aachen University
- 2010 - 2014 Coordinator of the large-scale collaborative research project “SYNFLOW” within the 7th Framework Program of the EU
- Since 2007 Scientific Director of CAT, the joint Catalytic Center of RWTH Aachen and Covestro
- 2002 - 2006 Coordinator of the BMBF funded ConNeCat lighthouse project “Regulated Systems for Multiphase Catalysis”
Professional Activities and Societies
- Since 2021 Advisory Editor of the journal “Angewandte Chemie”
- Since 2020 Vice-Chairman of the Board of DECHEMA, the German Society for Chemical Engineering and Biotechnology
- Since 2019 Review Panel of the Swiss National Competence Center (NCCR) in Catalysis
- 2018 - 2021 Editorial Board (Kuratorium) of the journal “Angewandte Chemie”
- 2018 - 2020 Advisory Board of the International Green Technology Association, Beijing, China
- Since 2015 Scientific Council of the German Society for Petroleum and Coal Science and Technology, DGMK
- 2014 - 2018 Scientific Advisory Board of the Renewable Energy Program of the Karlsruhe Institute of Technology KIT
- Since 2013 External Advisory Board of the UK Catalysis Hub
- Since 2013 Advisory Board of the Centre of Sustainable and Circular Technologies, Univ. of Bath, UK
- 2011 - 2016 Board of DECHEMA
- 2011 - 2014 Chairman of the German Catalysis Society GeCatS
- Since 2011 Honorary Member of the Chemical Society of Ethiopia
- Since 2010 Fellow of the Royal Society of Chemistry (FRSC)
- Since 2009 Scientific Advisory Board of the Center for Sustainable and Circular Technologies at the University of Bath
- 2009 - 2017 Strategy Council of RWTH Aachen University
- 2006 - 2014 Board of the Section “Sustainable Chemistry” of the German Chemical Society (GDCh)
- 2006 - 2008 Chairman of the Catalysis Section of DECHEMA
- 2005 - 2011 Board of the Petrochemical Division of the German Society for Petroleum and Coal Science and Technology (DGMK)
- Since 2005 Advisory Board of the journal “Advanced Synthesis and Catalysis”
- 2004 - 2016 Scientific Editor/Chairman of the Editorial Board of the journal “Green Chemistry”
Abteilungsmitglieder
PhD Studierende
Sihana AhmediMaurice Belleflamme
Himani Bisht
Ines Blaha (Gast)
Florian Dobreff
Manisha Durai
Furkan Düzenli
David Ezenarro
Hooman Ghazi Zahedi
Frederike Sophie Heinen
Junhui Hu
Zeynep Kap Dinler
Vishrant Kumar
Nitu Kumari
Arjit Mahana
Natalia Mendoza Vallejo
Markella Aliki Mermigki
Sebastian Obst
Edwin Onoh
Paul Resch
Angshuman Sarmah
Ilamparithy Selvakumar
Sebastian Stahl
Lisa Steinwachs
Hannah Stieber
Brayan Steven Torres-Barón
Gruppen
Forschungsprofil 'Molekulare Katalyse'
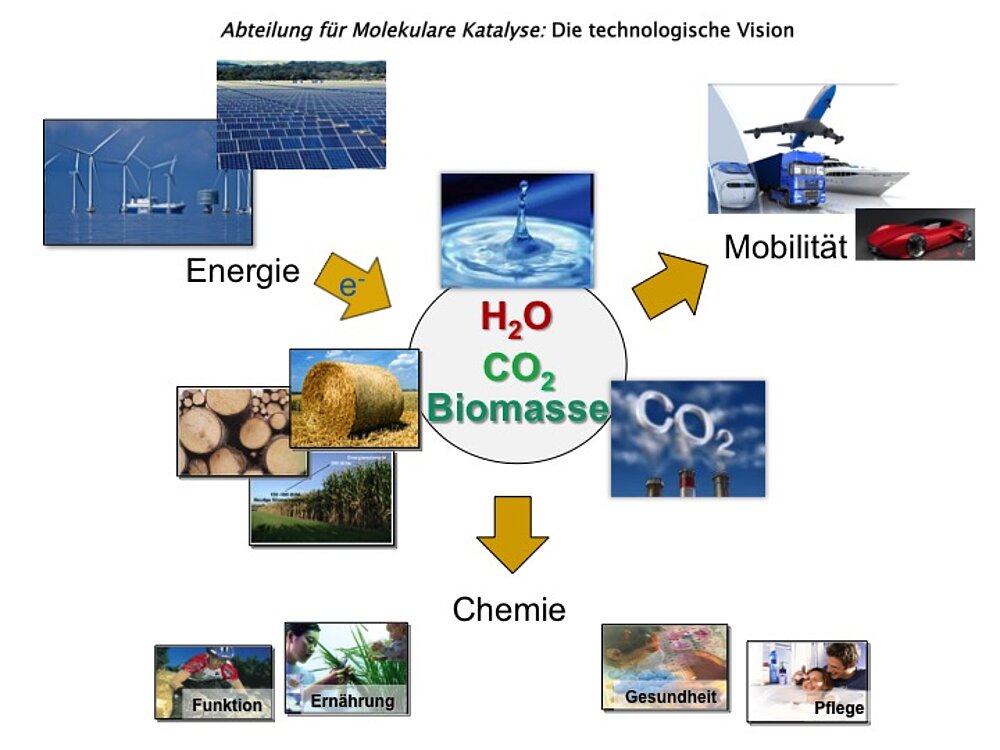
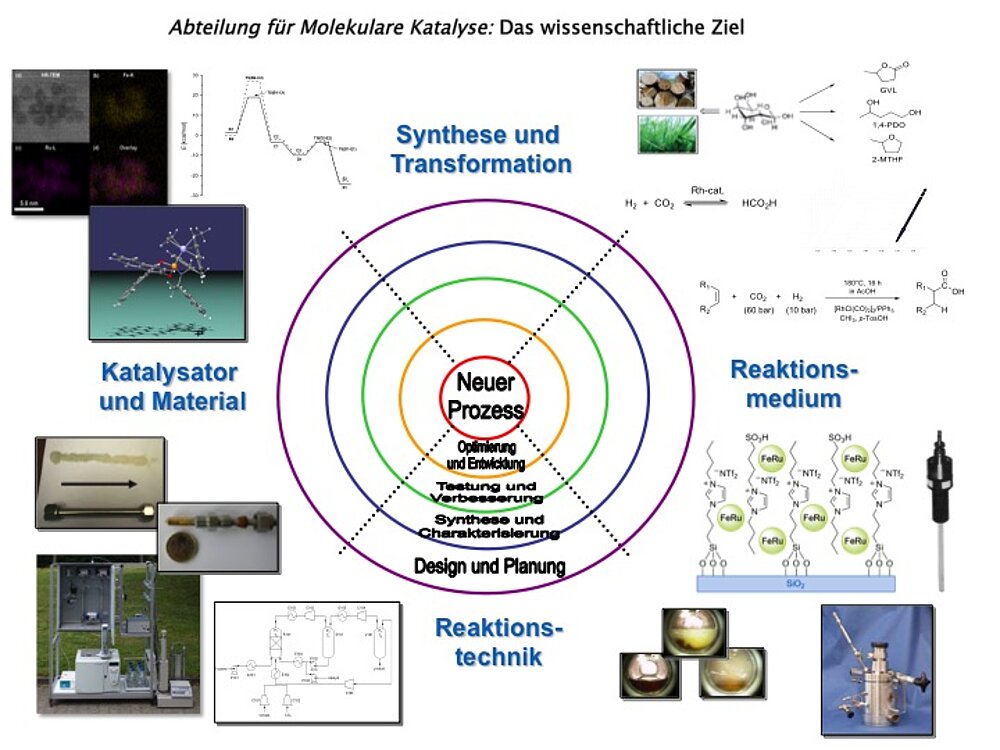
Die Grundlagenforschung der Abteilung 'Molekulare Katalyse' am MPI CEC leistet einen Beitrag zur langfristigen Entwicklung von Technologien, mit denen erneuerbare Energie und Rohstoffe als Grundlage für die Herstellung von chemischen Produkten und Kraftstoffen genutzt werden können.
Die wissenschaftlichen Fragestellungen fokussieren auf das Verständnis der molekularen Prinzipien der Katalyse als Basis für die skalen-übergreifende Beherrschung von katalytischen Prozessen an der Schnittstelle von Energie und Chemie.
Die wissenschaftlichen Fragestellungen der Abteilung „Molekulare Katalyse“ liegen auf dem Gebiet der metallorganischen Katalyse von ihren molekularen Grundlagen bis zur Entwicklung neuer reaktionstechnischer Konzepte. Die an den Prinzipien der „Green Chemistry“ ausgerichteten Forschungsthemen umfassen insbesondere die Nutzung von CO2 als C1-Baustein und die selektive Umwandlung biobasierter Rohstoffe, sowie den Einsatz alternativer Reaktionsmedien wie ionische Flüssigkeiten und überkritische Fluide. Die konkreten Konzepte für die Projektarbeit basieren auf einem grundlegenden Verständnis der zugrundeliegenden Mechanismen, wobei computer-gestützte Methoden eine wichtige Rolle für das rationale Katalysator-Design spielen.
Mögliche Anwendungen der Forschungsergebnisse reichen von der Produktion maßgeschneiderter Kraftstoffen, über die Herstellung von chemischen Zwischenprodukten bis zur Synthese von Feinchemikalien und Pharmazeutika. Die Forschungsarbeiten sind häufig in interdisziplinäre und internationale Kooperationen eingebunden, die sowohl erkenntnis-orientierte Forschungscluster als auch anwendungs-orientierte Technologieprojekte beinhalten.
Assoziierte Gruppen
Technikum - MPI für Kohlenforschung
Gruppenleiter: Dr. Nils Theyssen
CAT Catalytic Center Aachen - RWTH Aachen
Gruppenleiter: Dr. Veronica Grüntzig
Katalysatorimmobilisierung und Asymmetrische Katalyse - RWTH Aachen
Gruppenleiter: Dr. Giancarlo Franciò
Computergestütze Methoden in der Katalyse - RWTH Aachen
Gruppenleiter: Dr. Markus Hölscher